A quick peek into the report
Table of Contents
1.1 Product Definition
1.1.1 Product Definition
1.1.2 Inclusion and Exclusion Criteria
1.2 Market Scope
1.2.1 Scope of the Work
1.2.2 Key Questions Answered in the Report
1.3 Research Methodology
1.3.1 Global Cell and Gene Therapy Manufacturing QC Market
1.3.2 Data Sources
1.3.2.1 Primary Data Sources
1.3.2.2 Secondary Data Sources
1.3.3 Market Estimation Model
1.3.4 Criteria for Company Profiling
1.4 Market Overview
1.4.1 Global Market Scenario
1.4.1.1 Realistic Growth Scenario
1.4.1.2 Optimistic Scenario
1.4.1.3 Pessimistic Scenario
1.4.2 Market Footprint and Growth Potential
1.4.3 Future Potential
1.4.4 COVID-19 Impact on Market
1.4.4.1 Impact on Research and Clinical Operations
1.4.4.2 COVID-19 Impact: Current Scenario of the Market
1.4.4.3 Pre- and Post-COVID-19 Impact Assessment
1.4.4.3.1 Pre-COVID-19 Phase
1.4.4.3.2 Post-COVID-19 Phase
2.1 Regulatory Framework
2.1.1 Chemistry, Manufacturing, and Control (CMC) Requirements by the Food and Drug Administration (FDA)
2.1.1.1 Product Testing
2.1.1.2 Microbial Testing
2.1.1.3 Identity
2.1.1.4 Purity
2.1.1.5 Potency
2.1.1.6 Viability
2.1.1.7 Cell Number or Dose
2.1.2 Quality Aspects of Cell and Gene Therapy Products by the European Medicines Agency (EMA)
2.1.2.1 Characterization
2.1.2.2 Identity Testing
2.1.2.3 Purity Testing
2.1.3 Current Good Manufacturing Practice (CGMP) Regulations
2.1.3.1 U.S.
2.1.3.2 Europe
2.1.4 Global Regulatory Framework: Cell and Gene Therapy Manufacturing QC Market
3.1 Overview
3.2 Impact Analysis
3.3 Market Drivers
3.3.1 Growing Cell and Gene Therapy Production Leading to Increased Demand for Quality Control (QC)Testing
3.3.2 Increasing Number of Approvals Leading to an Upsurge in Demand for Cell and Gene Therapies QC Testing
3.3.3 Rise in Investment for the Development of Cell and Gene Therapies Increasing the Demand for QC Products and Services
3.4 Market Restraints
3.4.1 Limited Adoption of Cell and Gene Therapy due to High Manufacturing and QC Costs
3.5 Market Opportunities
3.5.1 Continuous Entry of New Market Participants in Cell and Gene Therapies to Create an Opportunity for the Expansion of Manufacturing Facilities and QC Testing Services
3.5.2 Introduction of Technologically Advanced Products in QC Testing for Cell and Gene Therapies
4.1 Overview
4.1 Key Strategies and Developments
4.1.1 Synergistic Activities
4.1.2 Product and Service Launches
4.1.3 Mergers and Acquisitions
4.1.4 Product Approvals
4.2 Market Share Analysis
4.3 Growth Share Analysis
4.3.1 Growth Share Analysis (by Application)
4.4 Supply Chain Analysis
4.4.1 Key Entities in Supply Chain
5.1 Overview
5.2 Cell Therapy
5.2.1 Autologous
5.2.2 Allogeneic
5.2.3 Others
5.3 Gene Therapy
5.3.1 Viral Vectors
5.3.2 Non-Viral Vectors
6.1 Overview
6.2 Products
6.2.1 Consumables
6.2.1.1 Kits and Assays
6.2.1.2 Reagents
6.2.1.3 Cell Culture Media and Supplements
6.2.2 Instruments
6.2.2.1 Bioreactors/Fermenters
6.2.2.2 PCR Systems
6.2.2.3 Chromatography System
6.2.2.4 Others
6.2.3 Software
6.3 Services
6.3.1 Safety Testing
6.3.2 Potency Testing
6.3.3 Identity Testing
6.3.4 Stability and Genetic Fidelity Testing
6.3.5 Others
7.1 Overview
7.2 Raw Material Preparation
7.3 Upstream Processing
7.4 Downstream Processing
7.5 Packaging
8.1 Overview
8.2 Polymerase Chain Reaction (PCR)
8.3 Flow Cytometry
8.4 Limulus Amebocyte Lysate (LAL)
8.5 Enzyme-Linked Immunosorbent Assay (ELISA)
8.6 Chromatography
8.7 Mass Spectrometry
8.8 Western Blotting
8.9 Next-Generation Sequencing (NGS)
8.10 Electrophoresis
8.11 Other Technologies
9.1 Overview
9.2 Safety Testing
9.2.1 By Safety Testing Type
9.3 Potency Testing
9.3.1 By Potency Testing Type
9.4 Identity Testing
9.4.1 By Identity Testing Type
9.5 Stability and Genetic Fidelity Testing
9.5.1 By Stability Testing and Genetic Fidelity Testing Type
9.6 Others
10.1 Overview
10.2 North America
10.2.1 North America Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.2.2 North America Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.2.3 North America Cell and Gene Therapy Manufacturing QC Market (by Process)
10.2.4 North America Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.2.5 North America Cell and Gene Therapy Manufacturing QC Market (by Application)
10.2.6 U.S.
10.2.6.1 U.S. Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.2.6.2 U.S. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.2.6.3 U.S. Cell and Gene Therapy Manufacturing QC Market (by Process)
10.2.6.4 U.S. Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.2.6.5 U.S. Cell and Gene Therapy Manufacturing QC Market (by Application)
10.2.7 Canada
10.2.7.1 Canada Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.2.7.2 Canada Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.2.7.3 Canada Cell and Gene Therapy Manufacturing QC Market (by Process)
10.2.7.4 Canada Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.2.7.5 Canada Cell and Gene Therapy Manufacturing QC Market (by Application)
10.3 Europe
10.3.1.1 Europe Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.3.1.2 Europe Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.3.1.3 Europe Cell and Gene Therapy Manufacturing QC Market (by Process)
10.3.1.4 Europe Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.3.1.5 Europe Cell and Gene Therapy Manufacturing QC Market (by Application)
10.3.2 U.K.
10.3.2.1 U.K. Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.3.2.2 U.K. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.3.2.3 U.K. Cell and Gene Therapy Manufacturing QC Market (by Process)
10.3.2.4 U.K. Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.3.2.5 U.K. Cell and Gene Therapy Manufacturing QC Market (by Application)
10.3.3 Germany
10.3.3.1 Germany Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.3.3.2 Germany Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.3.3.3 Germany Cell and Gene Therapy Manufacturing QC Market (by Process)
10.3.3.4 Germany Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.3.3.5 Germany Cell and Gene Therapy Manufacturing QC Market (by Application)
10.3.4 France
10.3.4.1 France Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.3.4.2 France Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.3.4.3 France Cell and Gene Therapy Manufacturing QC Market (by Process)
10.3.4.4 France Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.3.4.5 France Cell and Gene Therapy Manufacturing QC Market (by Application)
10.3.5 Italy
10.3.5.1 Italy Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.3.5.2 Italy Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.3.5.3 Italy Cell and Gene Therapy Manufacturing QC Market (by Process)
10.3.5.4 Italy Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.3.5.5 Italy Cell and Gene Therapy Manufacturing QC Market (by Application)
10.3.6 Spain
10.3.6.1 Spain Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.3.6.2 Spain Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.3.6.3 Spain Cell and Gene Therapy Manufacturing QC Market (by Process)
10.3.6.4 Spain Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.3.6.5 Spain Cell and Gene Therapy Manufacturing QC Market (by Application)
10.3.7 Rest-of-Europe
10.3.7.1 Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.3.7.2 Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.3.7.3 Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Process)
10.3.7.4 Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.3.7.5 Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Application)
10.4 Asia-Pacific
10.4.1.1 Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.4.1.2 Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.4.1.3 Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Process)
10.4.1.4 Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.4.1.5 Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Application)
10.4.2 China
10.4.2.1 China Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.4.2.2 China Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.4.2.3 China Cell and Gene Therapy Manufacturing QC Market (by Process)
10.4.2.4 China Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.4.2.5 China Cell and Gene Therapy Manufacturing QC Market (by Application)
10.4.3 Japan
10.4.3.1 Japan Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.4.3.2 Japan Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.4.3.3 Japan Cell and Gene Therapy Manufacturing QC Market (by Process)
10.4.3.4 Japan Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.4.3.5 Japan Cell and Gene Therapy Manufacturing QC Market (by Application)
10.4.4 South Korea
10.4.4.1 South Korea Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.4.4.2 South Korea Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.4.4.3 South Korea Cell and Gene Therapy Manufacturing QC Market (by Process)
10.4.4.4 South Korea Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.4.4.5 South Korea Cell and Gene Therapy Manufacturing QC Market (by Application)
10.4.5 Australia
10.4.5.1 Australia Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.4.5.2 Australia Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.4.5.3 Australia Cell and Gene Therapy Manufacturing QC Market (by Process)
10.4.5.4 Australia Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.4.5.5 Australia Cell and Gene Therapy Manufacturing QC Market (by Application)
10.4.6 India
10.4.6.1 India Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.4.6.2 India Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.4.6.3 India Cell and Gene Therapy Manufacturing QC Market (by Process)
10.4.6.4 India Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.4.6.5 India Cell and Gene Therapy Manufacturing QC Market (by Application)
10.4.7 Rest-of-Asia-Pacific
10.4.7.1 Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.4.7.2 Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.4.7.3 Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Process)
10.4.7.4 Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.4.7.5 Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Application)
10.5 Middle East and Africa
10.5.1.1 Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.5.1.2 Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.5.1.3 Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Process)
10.5.1.4 Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.5.1.5 Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Application)
10.5.2 Israel
10.5.2.1 Israel Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.5.2.2 Israel Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.5.2.3 Israel Cell and Gene Therapy Manufacturing QC Market (by Process)
10.5.2.4 Israel Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.5.2.5 Israel Cell and Gene Therapy Manufacturing QC Market (by Application)
10.5.3 Türkiye
10.5.3.1 Türkiye Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.5.3.2 Türkiye Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.5.3.3 Türkiye Cell and Gene Therapy Manufacturing QC Market (by Process)
10.5.3.4 Türkiye Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.5.3.5 Türkiye Cell and Gene Therapy Manufacturing QC Market (by Application)
10.5.4 K.S.A.
10.5.4.1 K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.5.4.2 K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.5.4.3 K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Process)
10.5.4.4 K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.5.4.5 K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Application)
10.5.5 U.A.E.
10.5.5.1 U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.5.5.2 U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.5.5.3 U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Process)
10.5.5.4 U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.5.5.5 U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Application)
10.5.6 South Africa
10.5.6.1 South Africa Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.5.6.2 South Africa Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.5.6.3 South Africa Cell and Gene Therapy Manufacturing QC Market (by Process)
10.5.6.4 South Africa Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.5.6.5 South Africa Cell and Gene Therapy Manufacturing QC Market (by Application)
10.5.7 Rest-of-Middle East and Africa
10.5.7.1 Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.5.7.2 Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.5.7.3 Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Process)
10.5.7.4 Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.5.7.5 Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Application)
10.6 Latin America
10.6.1.1 Latin America Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.6.1.2 Latin America Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.6.1.3 Latin America Cell and Gene Therapy Manufacturing QC Market (by Process)
10.6.1.4 Latin America Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.6.1.5 Latin America Cell and Gene Therapy Manufacturing QC Market (by Application)
10.6.2 Brazil
10.6.2.1 Brazil Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.6.2.2 Brazil Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.6.2.3 Brazil Cell and Gene Therapy Manufacturing QC Market (by Process)
10.6.2.4 Brazil Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.6.2.5 Brazil Cell and Gene Therapy Manufacturing QC Market (by Application)
10.6.3 Mexico
10.6.3.1 Mexico Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.6.3.2 Mexico Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.6.3.3 Mexico Cell and Gene Therapy Manufacturing QC Market (by Process)
10.6.3.4 Mexico Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.6.3.5 Mexico Cell and Gene Therapy Manufacturing QC Market (by Application)
10.6.4 Rest-of-Latin America
10.6.4.1 Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Offering)
10.6.4.2 Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
10.6.4.3 Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Process)
10.6.4.4 Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Technology)
10.6.4.5 Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Application)
11.1 Overview
11.2 Manufacturers
11.2.1 Bio-Techne Corporation
11.2.1.1 Company Overview
11.2.1.2 Role of Bio-Techne Corporation in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.1.3 Key Competitors
11.2.1.4 Financials
11.2.1.5 Analyst Perspective
11.2.2 bioMérieux SA
11.2.2.1 Company Overview
11.2.2.2 Role of bioMérieux SA in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.2.3 Key Competitors
11.2.2.4 Financials
11.2.2.5 Analyst Perspective
11.2.3 Danaher. (Cytiva)
11.2.3.1 Company Overview
11.2.3.2 Role of Danaher. (Cytiva) in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.3.3 Key Competitors
11.2.3.4 Financials
11.2.3.5 Key Insights about the Financial Health of the Company
11.2.3.5.1 Analyst Perspective
11.2.4 F. Hoffmann-La Roche Ltd
11.2.4.1 Company Overview
11.2.4.2 Role of F. Hoffmann-La Roche Ltd in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.4.3 Key Competitors
11.2.4.4 Financials
11.2.4.5 Key Insights about the Financial Health of the Company
11.2.4.6 Analyst Perspective
11.2.5 Lonza.
11.2.5.1 Company Overview
11.2.5.2 Role of Lonza. in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.5.3 Key Competitors
11.2.5.4 Financials
11.2.5.5 Analyst Perspective
11.2.6 Miltenyi Biotec B.V. & Co. KG
11.2.6.1 Company Overview
11.2.6.2 Role of Miltenyi Biotec B.V. & Co. KG in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.6.3 Key Competitors
11.2.6.4 Analyst Perspective
11.2.7 Sartorius AG
11.2.7.1 Company Overview
11.2.7.2 Role of Sartorius AG in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.7.3 Key Competitors
11.2.7.4 Financials
11.2.7.5 Analyst Perspective
11.2.8 Thermo Fisher Scientific Inc.
11.2.8.1 Company Overview
11.2.8.2 Role of Thermo Fisher Scientific Inc. in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.8.3 Key Competitors
11.2.8.4 Financials
11.2.8.5 Key Insights about the Financial Health of the Company
11.2.8.5.1 Analyst Perspective
11.2.9 WuXi AppTec
11.2.9.1 Company Overview
11.2.9.2 Role of WuXi AppTec in the Global Cell and Gene Therapy Manufacturing QC Market
11.2.9.3 Key Competitors
11.2.9.4 Financials
11.2.9.5 Analyst Perspective
11.3 Service
11.3.1 AGC Biologics.
11.3.1.1 Company Overview
11.3.1.2 Role of AGC Biologics. in the Global Cell and Gene Therapy Manufacturing QC Market
11.3.1.3 Key Competitors
11.3.1.4 Analyst Perspective
11.3.2 Charles River Laboratories International, Inc.
11.3.2.1 Company Overview
11.3.2.2 Role of Charles River Laboratories International, Inc. in the Global Cell and Gene Therapy Manufacturing QC Market
11.3.2.3 Key Competitors
11.3.2.4 Financials
11.3.2.5 Analyst Perspective
11.3.3 Catalent, Inc
11.3.3.1 Company Overview
11.3.3.2 Role of Catalent, Inc in the Global Cell and Gene Therapy Manufacturing QC Market
11.3.3.3 Key Competitors
11.3.3.4 Financials
11.3.3.5 Analyst Perspective
11.3.4 Eurofins Scientific
11.3.4.1 Company Overview
11.3.4.2 Role of Eurofins Scientific in the Global Cell and Gene Therapy Manufacturing QC Market
11.3.4.3 Key Competitors
11.3.4.4 Financials
11.3.4.5 Analyst Perspective
11.3.5 Merck KGaA
11.3.5.1 Company Overview
11.3.5.2 Role of Merck KGaA in the Global Cell and Gene Therapy Manufacturing QC Market
11.3.5.3 Key Competitors
11.3.5.4 Financials
11.3.5.5 Analyst Perspective
11.4 Emerging Companies
11.4.1 Applied StemCell, Inc.
11.4.2 Vineti
Table 1: Key Questions Answered in the Report
Table 2: Global Regulatory Scenario: Cell and Gene Therapy Manufacturing QC Market
Table 3: Impact Analysis, Market Drivers
Table 4: Investments in Cell and Gene Therapy Development, 2021-2022
Table 5: North America Cell and Gene Therapy Manufacturing QC Market, Impact Analysis
Table 6: Europe Cell and Gene Therapy Manufacturing QC Market Dynamics, Impact Analysis
Table 7: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Dynamics, Impact Analysis
Table 8: Approved Cell and Gene Therapies, Japan
Table 9: Latin America Cell and Gene Therapy Manufacturing QC Market Dynamics, Impact Analysis
Table 10: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market Dynamics, Impact Analysis
Figure 1: Cell and Gene Therapy Manufacturing QC Market, $Billion, 2022-2033
Figure 2: Global Cell and Gene Therapy Manufacturing QC Market, Impact Analysis
Figure 3: Global Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), % Share, 2022 and 2033
Figure 4: Global Cell and Gene Therapy Manufacturing QC Market (by Offering), % Share, 2022 and 2033
Figure 5: Global Cell and Gene Therapy Manufacturing QC Market (by Process), % Share, 2022 and 2033
Figure 6: Global Cell and Gene Therapy Manufacturing QC Market (by Technology), % Share, 2022 and 2033
Figure 7: Global Cell and Gene Therapy Manufacturing QC Market (by Application), % Share, 2022 and 2033
Figure 8: Global Cell and Gene Therapy Manufacturing QC Market (by Region), Market Snapshot
Figure 9: Global Cell and Gene Therapy Manufacturing QC Market Segmentation
Figure 10: Global Cell and Gene Therapy Manufacturing QC Market: Research Methodology
Figure 11: Primary Research Methodology
Figure 12: Bottom-Up Approach (Segment-Wise Analysis)
Figure 13: Top-Down Approach (Segment-Wise Analysis)
Figure 14: Global Cell and Gene Therapy Manufacturing QC Market Size and Growth Potential (Realistic Scenario), $Billion, 2022-2033
Figure 15: Global Cell and Gene Therapy Manufacturing QC Market Size and Growth Potential (Optimistic Scenario), $Billion, 2022-2033
Figure 16: Global Cell and Gene Therapy Manufacturing QC Market Size and Growth Potential (Pessimistic Scenario), $Billion, 2022-2033
Figure 17: Global Cell and Gene Therapy Manufacturing QC Market, $Billion, 2022-2033
Figure 18: Impact of COVID-19 on CGT Developmental Activities
Figure 19: Global Cell and Gene Therapy Manufacturing QC Market - Market Dynamics
Figure 20: U.S.FDA-Approved Cell and Gene Therapies, 2011-2022
Figure 21: Number of Cell and Gene Therapy Developers (by Region)
Figure 22: Share of Key Developments, January 2020-April 2023
Figure 23: Number of Product and Service Launches (by Company), January 2020 to April 2023
Figure 24: Market Share Analysis of the Global Cell and Gene Therapy Manufacturing QC Market (by Company), 2022
Figure 25: Growth Share Analysis for Global Cell and Gene Therapy Manufacturing QC Market (by Applications), 2022
Figure 26: Supply Chain Analysis
Figure 27: Global Cell and Gene Therapy Manufacturing QC Market (by Therapy Type)
Figure 28: Global Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), % Share, 2022 and 2033
Figure 29: Global Cell and Gene Therapy Manufacturing QC Market (Cell Therapy), $Billion, 2022-2033
Figure 30: Global Cell and Gene Therapy Manufacturing QC Market (Autologous), $Billion, 2022-2033
Figure 31: Global Cell and Gene Therapy Manufacturing QC Market (Allogeneic), $Billion, 2022-2033
Figure 32: Global Cell and Gene Therapy Manufacturing QC Market (Others), $Million, 2022-2033
Figure 33: Global Cell and Gene Therapy Manufacturing QC Market (Gene Therapy), $Billion, 2022-2033
Figure 34: Several Types of Viral Vectors Used in Gene Therapy
Figure 35: Global Cell and Gene Therapy Manufacturing QC Market (Viral Vectors), $Million, 2022-2033
Figure 36: Global Cell and Gene Therapy Manufacturing QC Market (Non-Viral Vectors), $Million, 2022-2033
Figure 37: Global Cell and Gene Therapy Manufacturing QC Market (by Offering)
Figure 38: Global Cell and Gene Therapy Manufacturing QC Market (by Offering), % Share, 2022 and 2033
Figure 39: Global Cell and Gene Therapy Manufacturing QC Market (Products), $Billion, 2022-2033
Figure 40: Global Cell and Gene Therapy Manufacturing QC Market (Products), % Share, 2022 and 2033
Figure 41: Global Cell and Gene Therapy Manufacturing QC Market (Consumables), $Million, 2022-2033
Figure 42: Global Cell and Gene Therapy Manufacturing QC Market (Kits and Assays), $Million, 2022-2033
Figure 43: Global Cell and Gene Therapy Manufacturing QC Market (Reagents), $Million, 2022-20333
Figure 44: Global Cell and Gene Therapy Manufacturing QC Market (Cell Culture Media), $Million, 2022-2033
Figure 45: Global Cell and Gene Therapy Manufacturing QC Market (Instruments), $Million, 2022-2033
Figure 46: Global Cell and Gene Therapy Manufacturing QC Market (Bioreactors/Fermenters), $Million, 2022-2033
Figure 47: Global Cell and Gene Therapy Manufacturing QC Market (PCR Systems), $Million, 2022-2033
Figure 48: Global Cell and Gene Therapy Manufacturing QC Market (Chromatography System), $Million, 2022-2033
Figure 49: Global Cell and Gene Therapy Manufacturing QC Market (Others), $Million, 2022-2033
Figure 50: Global Cell and Gene Therapy Manufacturing QC Market (Software), $Million, 2022-2033
Figure 51: Global Cell and Gene Therapy Manufacturing QC Market (Services), $Billion, 2022-2033
Figure 52: Global Cell and Gene Therapy Manufacturing QC Market (Services), % Share, 2022 and 2033
Figure 53: Global Cell and Gene Therapy Manufacturing QC Market (Safety Testing), $ Billion, 2022-2033
Figure 54: Global Cell and Gene Therapy Manufacturing QC Market (Potency Testing), $Million, 2022-2033
Figure 55: Global Cell and Gene Therapy Manufacturing QC Market (Identity Testing), $Million, 2022-2033
Figure 56: Global Cell and Gene Therapy Manufacturing QC Market (Stability and Genetic Fidelity Testing), $Million, 2022-2033
Figure 57: Global Cell and Gene Therapy Manufacturing QC Market (Others), $Million, 2022-2033
Figure 58: Global Cell and Gene Therapy Manufacturing QC Market (by Process)
Figure 59: Global Cell and Gene Therapy Manufacturing QC Market (by Process), % Share, 2022-2033
Figure 60: Global Cell and Gene Therapy Manufacturing QC Market (Raw Material Preparation), $Million, 2022-2033
Figure 61: Followed Steps in Upstream Processing
Figure 62: Global Cell and Gene Therapy Manufacturing QC Market (Upstream Processing), $Billion, 2022-2033
Figure 63: Followed Steps in Downstream Processing
Figure 64: Global Cell and Gene Therapy Manufacturing QC Market (Downstream Processing), $Billion, 2022-2033
Figure 65: Steps Involved in Packaging Operations
Figure 66: Global Cell and Gene Therapy Manufacturing QC Market (Packaging), $Million, 2022-2033
Figure 67: Global Cell and Gene Therapy Manufacturing QC Market (by Technology)
Figure 68: Global Cell and Gene Therapy Manufacturing QC Market (by Technology), % Share, 2022 and 2033
Figure 69: Global Cell and Gene Therapy Manufacturing QC Market (PCR), $Million, 2022-2033
Figure 70: Global Cell and Gene Therapy Manufacturing QC Market (Flow Cytometry), $Million,2022-2033
Figure 71: Global Cell and Gene Therapy Manufacturing QC Market (LAL), $Million,2022-2033
Figure 72: Global Cell and Gene Therapy Manufacturing QC Market (ELISA), $Million, 2022-2033
Figure 73: Global Cell and Gene Therapy Manufacturing QC Market (Chromatography), $Million, 2022-2033
Figure 74: Global Cell and Gene Therapy Manufacturing QC Market (Mass Spectrometry), $Million, 2022-2033
Figure 75: Global Cell and Gene Therapy Manufacturing QC Market (Western Blotting), $Million, 2022-2033
Figure 76: Global Cell and Gene Therapy Manufacturing QC Market (NGS), $Million, 2022-2033
Figure 77: Global Cell and Gene Therapy Manufacturing QC Market (Electrophoresis), $Million, 2022-2033
Figure 78: Global Cell and Gene Therapy Manufacturing QC Market (Other Technologies), $Million, 2022-2033
Figure 79: Global Cell and Gene Therapy Manufacturing QC Market (by Application)
Figure 80: Global Cell and Gene Therapy Manufacturing QC Market (by Application), % Share, 2022 and 2033
Figure 81: Global Cell and Gene Therapy Manufacturing QC Market (Safety Testing), $Billion, 2022-2033
Figure 82: Global Cell and Gene Therapy Manufacturing QC Market (by Safety Testing Type), % Share, 2022 and 2033
Figure 83: Global Cell and Gene Therapy Manufacturing QC Market (Potency Testing), $Million, 2022-2033
Figure 84: Global Cell and Gene Therapy Manufacturing QC Market (by Potency Testing Type), % Share, 2022 and 2033
Figure 85: Global Cell and Gene Therapy Manufacturing QC Market (Identity Testing), $Million, 2022-2033
Figure 86: Global Cell and Gene Therapy Manufacturing QC Market (by Identity Testing Type), % Share, 2022 and 2033
Figure 87: Global Cell and Gene Therapy Manufacturing QC Market (Stability and Genetic Fidelity Testing), $Million, 2022-2033
Figure 88: Global Cell and Gene Therapy Manufacturing QC Market (by Identity Testing Type), % Share, 2022 and 2033
Figure 89: Global Cell and Gene Therapy Manufacturing QC Market (Others), $Million, 2022-2033
Figure 90: Global Cell and Gene Therapy Manufacturing QC Market (by Region), Market Snapshot
Figure 91: North America Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 92: North America Cell and Gene Therapy Manufacturing QC Market (by Country), % Share, 2022 and 2033
Figure 93: North America Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 94: North America Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 95: North America Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 96: North America Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 97: North America Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 98: U.S. Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 99: U.S. Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 100: U.S. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 101: U.S. Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 102: U.S. Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 103: U.S. Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 104: Canada Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 105: Canada Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 106: Canada Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 107: Canada Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 108: Canada Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 109: Canada Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 110: Europe Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 111: Europe Cell and Gene Therapy Manufacturing QC Market (by Country), % Share, 2022-2033
Figure 112: Europe Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 113: Europe Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 114: Europe Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 115: Europe Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 116: Europe Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 117: U.K. Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 118: U.K. Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 119: U.K. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 120: U.K. Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 121: U.K. Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 122: U.K. Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 123: Germany Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 124: Germany Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 125: Germany Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 126: Germany Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 127: Germany Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 128: Germany Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 129: France Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 130: France Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 131: France Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 132: France Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 133: France Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 134: France Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 135: Italy Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 136: Italy Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 137: Italy Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 138: Italy Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 139: Italy Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 140: Italy Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 141: Spain Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 142: Spain Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 143: Spain Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 144: Spain Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 145: Spain Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 146: Spain Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 147: Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 148: Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 149: Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 150: Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 151: Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 152: Rest-of-Europe Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 153: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 154: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Country), % Share, 2022-2033
Figure 155: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 156: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 157: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 158: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 159: Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 160: China Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 161: China Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 162: China Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 163: China Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 164: China Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 165: China Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 166: Japan Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 167: Japan Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 168: Japan Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 169: Japan Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 170: Japan Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 171: Japan Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 172: South Korea Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 173: South Korea Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 174: South Korea Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 175: South Korea Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 176: South Korea Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 177: South Korea Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 178: Australia Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 179: Australia Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 180: Australia Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 181: Australia Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 182: Australia Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 183: Australia Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 184: India Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 185: India Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 186: India Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 187: India Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 188: India Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 189: India Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 190: Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 191: Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 192: Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 193: Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 194: Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 195: Rest-of-Asia-Pacific Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 196: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 197: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Country), % Share, 2022-2033
Figure 198: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 199: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 200: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 201: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 202: Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 203: Israel Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 204: Israel Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 205: Israel Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 206: Israel Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 207: Israel Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 208: Israel Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 209: Türkiye Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 210: Türkiye Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 211: Türkiye Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 212: Türkiye Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 213: Türkiye Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 214: Türkiye Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 215: K.S.A. Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 216: K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 217: K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 218: K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 219: K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 220: K.S.A. Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 221: U.A.E. Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 222: U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 223: U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 224: U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 225: U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 226: U.A.E. Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 227: South Africa Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 228: South Africa Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 229: South Africa Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 230: South Africa Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 231: South Africa Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 232: South Africa Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 233: Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 234: Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 235: Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 236: Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 237: Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 238: Rest-of-Middle East and Africa Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 239: Latin America Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 240: Latin America Cell and Gene Therapy Manufacturing QC Market (by Country), % Share, 2022-2033
Figure 241: Latin America Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 242: Latin America Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 243: Latin America Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 244: Latin America Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 245: Latin America Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 246: Brazil Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 247: Brazil Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 248: Brazil Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 249: Brazil Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 250: Brazil Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 251: Brazil Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 252: Mexico Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 253: Mexico Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 254: Mexico Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 255: Mexico Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 256: Mexico Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 257: Mexico Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 258: Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market, $Million, 2022-2033
Figure 259: Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Offering), $Million, 2022-2033
Figure 260: Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Therapy Type), $Million, 2022-2033
Figure 261: Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Process), $Million, 2022-2033
Figure 262: Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Technology), $Million, 2022-2033
Figure 263: Rest-of-Latin America Cell and Gene Therapy Manufacturing QC Market (by Application), $Million, 2022-2033
Figure 264: Total Number of Companies Profiled
Figure 265: Bio-Techne Corporation: Product Portfolio
Figure 266: Bio-Techne Corporation: Overall Financials, $Million, 2020-2022
Figure 267: Bio-Techne Corporation: Revenue (by Segment), $Million, 2020-2022
Figure 268: Bio-Techne Corporation: Revenue (by Region), $Million, 2020-2022
Figure 269: bioMérieux SA: Product Portfolio
Figure 270: bioMérieux SA: Overall Financials, $Million, 2020-2022
Figure 271: bioMérieux SA: Revenue (by Segment), $Million, 2020-2022
Figure 272: bioMérieux SA: Revenue (by Region), $Million, 2020-2022
Figure 273: Danaher. (Cytiva): Product Portfolio
Figure 274: Danaher. (Cytiva): Overall Financials, $Million, 2020-2022
Figure 275: Danaher. (Cytiva): Revenue (by Segment), $Million, 2020-2022
Figure 276: Danaher. (Cytiva): Revenue (by Region), $Million, 2020-2022
Figure 277: Danaher. (Cytiva): R&D Expenditure, $Million, 2019-2021
Figure 278: F. Hoffmann-La Roche Ltd: Product Portfolio
Figure 279: F. Hoffmann-La Roche Ltd: Overall Financials, $Million, 2020-2022
Figure 280: F. Hoffmann-La Roche Ltd: Revenue (by Segment), $Million, 2020-2022
Figure 281: F. Hoffmann-La Roche Ltd: Revenue (by Region), $Million, 2020-2022
Figure 282: F. Hoffmann-La Roche Ltd: R&D Expenditure, $Million, 2020-2022
Figure 283: Lonza.: Product Portfolio
Figure 284: Lonza.: Overall Financials, $Million, 2020-2022
Figure 285: Lonza.: Revenue (by Segment), $Million, 2021and 2022
Figure 286: Miltenyi Biotec B.V. & Co. KG: Product Portfolio
Figure 287: Sartorius AG: Product Portfolio
Figure 288: Sartorius AG: Overall Financials, $Million, 2020-2022
Figure 289: Sartorius AG: Revenue (by Segment), $Million, 2020-2022
Figure 290: Sartorius AG: Revenue (by Region), $Million, 2020-2022
Figure 291: Thermo Fisher Scientific Inc.: Product Portfolio
Figure 292: Thermo Fisher Scientific Inc.: Overall Financials, $Million, 2020-2022
Figure 293: Thermo Fisher Scientific Inc.: Revenue (by Segment), $Million, 2020-2022
Figure 294: Thermo Fisher Scientific Inc.: Revenue (by Region), $Million, 2020-2022
Figure 295: Thermo Fisher Scientific Inc.: R&D Expenditure, 2020-2022
Figure 296: WuXi AppTec: Product Portfolio
Figure 297: WuXi AppTec: Overall Financials, $Million, 2020-2022
Figure 298: WuXi AppTec: Revenue (by Segment), $Million, 2021 and 2022
Figure 299: AGC Biologics.: Product Portfolio
Figure 300: Charles River Laboratories International, Inc.: Product Portfolio
Figure 301: Charles River Laboratories International, Inc.: Overall Financials, $Million, 2020-2022
Figure 302: Charles River Laboratories International, Inc: Revenue (by Segment), $Million, 2020-2022
Figure 303: Charles River Laboratories International, Inc.: Revenue (by Region), $Million, 2020-2022
Figure 304: Catalent, Inc: Product Portfolio
Figure 305: Catalent, Inc: Overall Financials, $Million, 2020-2022
Figure 306: Catalent, Inc: Revenue (by Segment), $Million, 2020-2022
Figure 307: Catalent, Inc: Revenue (by Region), $Million, 2020-2022
Figure 308: Eurofins Scientific: Product Portfolio
Figure 309: Eurofins Scientific.: Overall Financials, $Million, 2020-2022
Figure 310: Eurofins Scientific.: Revenue (by Segment), $Million, 2020-2022
Figure 311: Eurofins Scientific.: Revenue (by Region), $Million, 2020-2022
Figure 312: Merck KGaA: Product Portfolio
Figure 313: Merck KGaA: Overall Financials, $Million, 2020-2022
Figure 314: Merck KGaA: Revenue (by Segment), $Million, 2020-2022
Figure 315: Merck KGaA: Revenue (by Region), $Million, 2020-2022
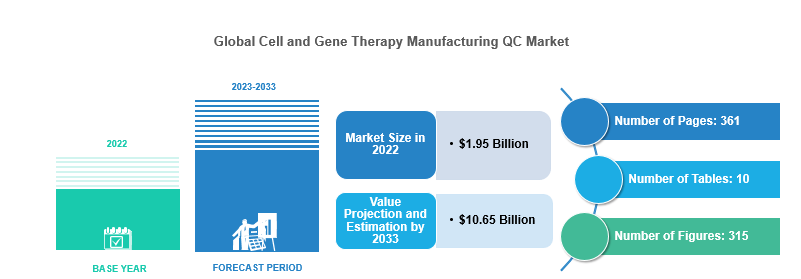
Market Report Coverage
Key Market Players and Competition Synopsis
Cell and gene-based therapies have sparked efforts to promote treatments such as chimeric antigen receptor (CAR)-T cell therapy has gained significant attention in the field of oncology for its potential to treat certain types of cancer. The need to cater to the clinical and commercial production of cell-based therapies has increased the demand for quality control (QC) testing during the manufacturing of cell and gene-based therapies. The increased demand for treatments in the healthcare sector is fuelling the expansion of the cell and gene therapy manufacturing QC market and helping the market to spread out across different regions.
Key Companies Profiled:
• Bio-Techne Corporation
• bioMérieux SA
• Danaher. (Cytiva)
• F. Hoffmann-La Roche Ltd
• Lonza.
• Miltenyi Biotec B.V. & Co. KG
• Sartorius AG
• Thermo Fisher Scientific Inc.
• WuXi AppTec
• AGC Biologics.
• Charles River Laboratories International, Inc.
• Catalent, Inc
• Eurofins Scientific
• Merck KGaA
How can this report add value to an organization?
Workflow/Innovation Strategy: The cell and gene therapy manufacturing QC market (by offering) has been segmented into products and services. Moreover, the study provides the reader with a detailed understanding of the different applications of cell and gene therapy manufacturing QC in raw material preparation, upstream processing, downstream processing, and packaging.
Growth/Marketing Strategy: Cell and gene therapy manufacturing QC is being used for raw material preparation, upstream processing, downstream processing, and packaging. Various companies are providing products and services to aid in the manufacturing and QC of various cell and gene therapies, which is also the key strategy for market players to excel in the current cell and gene therapy manufacturing QC market.
Competitive Strategy: Key players in the global cell and gene therapy manufacturing QC market have been analyzed and profiled in the study, including manufacturers involved in new product launches, acquisitions, expansions, and strategic collaborations. Moreover, a detailed competitive benchmarking of the players operating in the global cell and gene therapy manufacturing QC market has been done to help the reader understand how players stack against each other, presenting a clear market landscape. Additionally, comprehensive competitive strategies such as partnerships, agreements, and collaborations will aid the reader in understanding the untapped revenue pockets in the market.
Cell and Gene Therapy Manufacturing QC Industry and Technology Overview
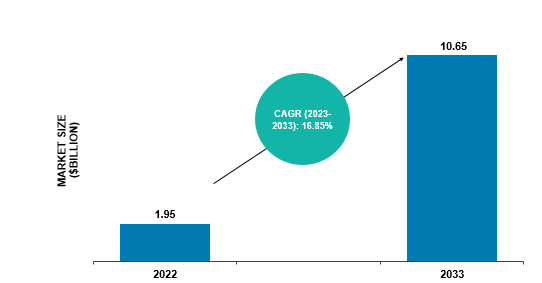
The global cell and gene therapy manufacturing QC market was valued at $1.95 billion in 2022 and is anticipated to reach $10.65 billion by 2033, witnessing a CAGR of 16.85% during the forecast period 2023-2033. The growth in the global cell and gene therapy manufacturing QC market is expected to be driven by the increased number of approved therapies and growing infrastructure requirements. In addition, expansion in target indications for cell and gene-based therapies creates a demand for large-scale manufacturing and QC.
Market Lifecycle Stage
The global cell and gene therapy manufacturing QC market is in progressing phase. The cell and gene therapy manufacturing QC market is experiencing rapid growth due to the increasing adoption of innovative therapies such as CAR T-cells and others. Robust quality control processes are essential to ensure the safety, efficacy, and consistency of cell and gene-based treatments. The FDA has approved more than 25 cell and gene-based therapies in the last 10 years. These factors are expected to drive the demand for consumables, instruments, and software solutions required for manufacturing cell and gene therapy, thereby augmenting the growth of the cell and gene therapy manufacturing QC market.
Figure 1: Global Cell and Gene Therapy Manufacturing QC Market Snapshot
Source: BIS Research Analysis
Impact
The field of medicine is transformed with the commercialization of cell and gene therapies. With the advent of time and introduction of new technologies, cell and gene therapy areas are flourishing. There is constant ongoing research for the development of novel cell and gene therapies. According to the American Society of Gene and Cell Therapy (ASGCT), as of February 2023, there are more than 2,000 clinical trials in the pipeline. The robust clinical pipeline for novel cell and gene entities is expected to create a lucrative opportunity for QC and boost the growth of the cell and gene therapy manufacturing QC market. In addition, the entry of several established players, such as Lonza., Merck KGaA, Charles River Laboratories International Inc., Eurofins Scientific, and others, is expected to aid the market growth.
The field of cell and gene therapy manufacturing QC is witnessing several trends and advancements that are expected to have a significant impact on the market. Some of the trends include the adoption of automation QC processes, advanced analytical technologies, process analytical testing (PAT) and quality risk management (QRM), and others. These trends and advancements in cell and gene therapy manufacturing QC are expected to drive improvements in product quality, manufacturing efficiency, regulatory compliance, and patient safety. They are expected to play a crucial role in supporting the growth and success of the cell and gene therapy industry, making QC an integral part of the development and commercialization of these advanced therapies.
Impact of COVID-19
In December 2019, Wuhan, a city in the Hubei region of China, was the site of the first detection of the COVID-19 outbreak. Following the classification of COVID-19 as novel pneumonia due to a cluster of unexplained pneumonia cases, efforts to pinpoint the culprit causing the outbreak and outline its genomic sequence got underway right once. The virus has already spread to every country on the globe, and researchers, governments, and business leaders are working to find answers to the crisis at a scale and speed that has never been seen. Testing for SARS-CoV-2 in the populace is one of the main steps that has been put into place globally, among many other measures used to stop the spread of the disease. The most crucial benefit of testing is that it offers evidence of illness, enabling those who are tested and those they have come into contact with to take the required precautions, including quarantining, to minimize community exposure.
The COVID-19 pandemic has substantially interrupted social, economic, and political activity around the world due to its unparalleled size and intensity. As a result, the cell and gene therapy (CGT) sector, which has historically struggled with tremendous complexity in the supply of materials, production, and logistical operations, has been disrupted by COVID-19.
The research, production, clinical development, and market introduction of cell and gene-based therapies (CGTs) for diseases unrelated to COVID-19 have all been significantly disrupted as a result of the COVID-19 pandemic. A lack of manufacturing material supplies, challenges with clinical studies, and a delay in the creation of regulatory dossiers are all significant reasons for the manufacturing of cell and gene therapy. This has emphasized the significance of tackling the difficulties in CGTs' supply chain and production to increase resilience during the crisis.
To prevent CGTs' market access from being significantly disrupted, manufacturing resilience, digitalization, telemedicine, value-based pricing, and creative payment systems may be progressively tapped.
Market Segmentation:
Segmentation 1: by Therapy Type
• Cell Therapy
• Gene Therapy
Based on therapy type, the cell therapy segment dominated the global cell and gene therapy manufacturing QC market in FY2022. The increasing adoption of cell-based therapies, such as CAR-T cell therapy and stem cell therapy, contributed to the prominence of the cell therapy segment. Cell therapy is a type of medical treatment that involves the transplantation or infusion of live, healthy cells into a patient's body to treat or cure a disease or disorder. The goal of cell therapy is to restore or replace damaged or malfunctioning cells with healthy ones or to use the therapeutic properties of the transplanted cells to stimulate the body's own natural healing processes.
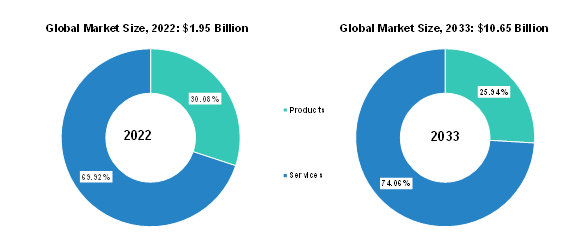
Segmentation 2: by Offering
• Products
• Services
Source: BIS Research Analysis
Based on offering, the services segment dominated the global cell and gene therapy manufacturing QC market in FY2022. This segment encompassed a range of vital services, including quality control testing, analytical services, process development, validation, and regulatory compliance. Within the services segment, there is a further division into various categories, including safety testing, potency testing, sterility testing, identity testing, stability and genetic fidelity testing, and others.
Segmentation 3: by Process
• Raw Material Preparation
• Upstream Processing
• Downstream Processing
• Packaging
Based on process, the global cell and gene therapy manufacturing QC market was dominated by the upstream processing segment in FY2022. The dominance of the upstream processing segment can be attributed to several factors, including advancements in bioprocessing technologies, optimized cell culture media, and improved bioreactor systems.
Segmentation 4: by Application
• Safety Testing
• Potency Testing
• Identity Testing
• Stability and Genetic Fidelity Testing
• Others
Based on application, the safety testing segment accounted for the largest share of the global cell and gene therapy manufacturing QC market in FY2022. This segment encompasses a range of tests, including sterility testing, endotoxin testing, and among others. Furthermore, some of the key players, such as Lonza., Thermo Fisher Scientific Inc., Charles River Laboratories International, Inc., Eurofins Scientific, and Merck KGaA, offer safety testing, potency testing, and other services in the global cell and gene therapy manufacturing QC market.
Segmentation 5: by Technology
• Polymerase Chain Reaction (PCR)
• Flow Cytometry
• Limulus Amebocyte Lysate (LAL)
• Enzyme-Linked Immunosorbent Assay (ELISA)
• Chromatography
• Mass Spectrometry
• Western Blotting
• Next-Generation Sequencing (NGS)
• Electrophoresis
• Other Technologies
Based on technology, the global cell and gene therapy manufacturing QC market is dominated by the PCR segment in FY2022. It plays a vital role in various QC (quality control) processes, including gene expression analysis, viral vector detection, and genetic stability assessment. PCR is used in a variety of tasks in cell and gene therapy manufacturing, including detecting contaminants, measuring gene expression and viral vector integration, and assessing gene editing success. Different types of PCR systems, such as real-time PCR and digital PCR, are used depending on the specific application and process requirements.
Segmentation 6: by Region
• North America
• Europe
• Asia-Pacific
• MEA
• LATAM
North America cell and gene therapy manufacturing QC market is expected to have the highest market share in 2022 and is currently the leading contributor to the market. However, the Asia-Pacific region, constituting several emerging economies, is expected to register the highest CAGR of 18.01% during the forecast period 2023-2033.
Recent Developments in the Global Cell and Gene Therapy Manufacturing QC Market
• In March 2023, Thermo Fisher Scientific Inc. collaborated with Arsenal Biosciences to assist in the clinical manufacturing of autologosus T-cell therapies.
This partnership combined Thermo Fisher's cell therapy manufacturing expertise with Arsenal Biosciences' innovative technologies, with the goal of advancing the development and accessibility of personalized T-cell therapies.
• In January 2023, Sartorius AG collaborated with Roosterbio Inc. to enhance their downstream purification methods in the field of exosome development.
• In February 2023, Charles River Laboratories International, Inc. partnered with Purespring Therapeutics. This partnership would advance gene therapies for kidney diseases and provide innovative treatment options for patients by leveraging Charles River's eXpDNA plasmid platform.
• In January 2023, Bio-Techne Corporation launched MauriceFlex, a new product under its ProteinSimple brand. MauriceFlex is a versatile system that facilitates protein charge variant fractionation along with routine cIEF (capillary isoelectric focusing) and CE-SDS (capillary electrophoresis-sodium dodecyl sulfate) assays. This innovative system offered a comprehensive solution for protein characterization, streamlining workflows in protein analysis.
• In January 2023, Bio-Techne Corporation launched RNAscope plus assay to advance its gene therapy development.
• In April 2023, Danaher. (Cytiva) launched X-platform bioreactors, which aim to streamline single-use upstream bioprocessing operations. These versatile bioreactors can be used for producing monoclonal antibodies, protein-based drugs, cell and gene therapies, and viral vectors. They offer flexibility and efficiency in bioprocessing, facilitating the development and manufacturing of various therapeutic products.
Demand - Drivers and Limitations
Market Demand Drivers:
• Growing Cell and Gene Therapy Production Leading to Increased Demand for Quality Control (QC)Testing
• Increasing Number of Approvals Leading to an Upsurge in Demand for Cell and Gene Therapies QC Testing
• Rise in Investment for the Development of Cell and Gene Therapies Increasing the Demand for QC Products and Services
Market Restraints:
• Limited Adoption of Cell and Gene Therapy due to High Manufacturing and QC Costs
Market Opportunities:
• Continuous Entry of New Market Participants in Cell and Gene Therapies to Create an Opportunity for the Expansion of Manufacturing Facilities and QC Testing Services
• Introduction of Technologically Advanced Products in QC Testing for Cell and Gene Therapies
Analyst View
According to Akash Mhaskar, Principal Analyst, BIS Research, “The global cell and gene therapy manufacturing QC market is experiencing rapid growth due to the increasing adoption of innovative therapies such as CAR T-cells and others. Robust quality control processes are essential to ensure the safety, efficacy, and consistency of cell and gene-based treatments. The market is driven by the need for stringent QC measures throughout the manufacturing journey, advancements in manufacturing technologies, and expanding regulatory frameworks. The global cell and gene therapy manufacturing QC market has witnessed a steady growth in the number of new entrants and investments in cell and gene therapy, thereby driving a surge in demand for the products and services required in the manufacturing and QC of cell and gene therapies. This trend is also expected to continue over the forecast period. Therefore, the impact of the aforementioned factors is expected to drive the global cell and gene therapy manufacturing QC market in the near future.”
Cell and Gene Therapy Manufacturing QC Market - A Global and Regional Analysis
Focus on Therapy Type, Offering, Process, Technology, Application, and Region - Analysis and Forecast, 2023-2033
Frequently Asked Questions
Cell and gene therapy manufacturing QC refers to the quality control processes and procedures implemented throughout the manufacturing and production of cell and gene therapies. It involves a set of measures and tests aimed at ensuring the safety, efficacy, and consistent quality of these advanced therapies. The QC procedures encompass various stages of manufacturing, including raw material testing, process validation, in-process monitoring, and final product testing.
New technologies and methods are rapidly emerging to enhance the manufacturing and delivery of new drugs, which is driving the market for cell and gene therapy manufacturing QC. Here are some prevalent patterns in the industry:
Increased Emphasis on Automation: As the market for cell and gene therapies expands, more effective and scalable production techniques are becoming increasingly important. Artificial intelligence and robotics are two automation technologies that are rapidly being used to streamline the manufacturing process, lower costs, and enhance quality control.
Production Capacity Expansion for Cell and Gene Therapies: To address the rising demand for these treatments, many businesses in the industry are expanding their manufacturing capabilities. Enhancing production quantities includes building new production plants and expanding current ones.
Adoption of Closed and Variable Manufacturing Systems: The market for cell and gene therapy manufacturing QC is seeing an increase in the adoption of closed and variable manufacturing systems. These systems are made to reduce the possibility of contamination and improve the effectiveness of the production process.
These are only a few illustrations of the major developments in the industry for cell and gene therapy manufacturing. Emerging innovations and technologies are anticipated to emerge as the area develops further, helping to influence the industry's future.
The global cell and gene therapy manufacturing QC market is currently witnessing several developments, primarily aimed toward bringing new products and services. Major manufacturers of cell and gene therapy manufacturing QC products, along with the service providers, are actively undertaking significant business strategies to translate success in research and development into the commercial clinical setting. Companies such as Sartorius AG, Merck KGaA, Lonza., Thermo Fisher Scientific Inc., and Bio-Rad Laboratories, Inc. have majorly adopted partnerships, collaborations, and joint venture strategies.
A new entrant can focus on various cell and gene therapy manufacturing QC product offerings by established players. The market is fragmented with established market leaders such as Sartorius AG, Merck KGaA, Lonza., Thermo Fisher Scientific Inc., and Bio-Rad Laboratories, Inc., who have had their presence in the market for the past many years. The new entrants can focus on their strategy of product launches and global expansions for cell and gene therapy manufacturing QC.
• The vast segmentation of the report
• Comparison of analytical methods that are offered by the companies as a service to the customers
• Supply chain analysis of cell and gene therapy manufacturing
• Competitive Benchmarking of key players for each of the key players of cell and gene therapy manufacturing QC
• Growth share analysis for key segments
• The report also comprises key regulations, competitive landscape, key drivers and restraints, patent analysis, business model analysis, and company profiles with a detailed analysis of the strategies undertaken by the major company
• Companies that manufacture and commercialize consumables and equipment required in the manufacturing QC of cell and gene therapy
• Any new entrant that wants to know where the opportunities lie in the cell and gene therapy manufacturing QC market
• Any established player that wants to expand its product portfolio or business in new geographies



