A quick peek into the report
Table of Contents
1.1 Inclusions and Exclusions
2.1 Scope of Work
2.2 Key Questions Answered in the Report
3.1 Global mRNA Vaccines and Therapeutics Market: Research Methodology
3.2 Primary Data Sources
3.3 Secondary Data Sources
3.4 Market Estimation Model
3.5 Criteria for Company Profiling
4.1 Market Definition
4.2 Current Landscape and Future Potential of mRNA-Based Vaccines
4.3 Market Footprint and Growth Potential
4.3.1 Global mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, 2020-2025
4.3.2 Global mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines), $Million, 2025-2031
5.1 Overview
5.2 Legal Requirements and Frameworks in the U.S.
5.2.1 Vaccines
5.2.1.1 Pre-Clinical and Clinical Development
5.2.1.2 Emergency Use Authorization (Public Health Emergencies)
5.2.1.3 Assessment of Manufacturing
5.2.1.4 Final Approval
5.2.2 Therapeutics
5.2.2.1 Clinical Trial Authorization
5.2.2.2 Marketing Authorization
5.2.2.3 Post-Authorization Regulations
5.3 Legal Requirements and Frameworks in Europe
5.3.1 Vaccines
5.3.2 Therapeutics
5.3.2.1 Centralized Procedure
5.3.2.2 Decentralized Procedure
5.3.2.3 Mutual-Recognition Procedure
5.3.2.4 National Procedure
5.4 Legal Requirements and Frameworks in Asia-Pacific
5.4.1 China
5.4.2 Japan
6.1 Overview
6.2 Impact Analysis
6.3 Market Drivers
6.3.1 Growing Cases of COVID-19 Globally
6.3.2 Favorable Regulatory Environment
6.3.3 Significant External Funding for the Development of mRNA-Based COVID-19 Vaccines
6.4 Market Restraints
6.4.1 Logistical Challenges Associated with mRNA-Based COVID-19 Vaccines
6.4.2 High Price Associated with mRNA-Based COVID-19 Vaccines
6.5 Opportunities
6.5.1 Growing Emphasis on Research and Development for Non-COVID-19 Applications
7.1 Key Strategies and Developments
7.1.1 Synergistic Activities
7.1.2 Product Approvals and Commercialization
7.1.3 Funding
7.2 Market Share Analysis (by Company), 2020-2021
8.1 COVID-19 mRNA Vaccines
8.1.1 Overview
8.1.2 Commercialized Vaccines
8.1.2.1 mRNA-1273
8.1.2.1.1 Supply Agreements for mRNA-1273
8.1.2.2 BNT162b2
8.1.2.2.1 Supply Agreements for BNT162b2 (COMIRNATY)
8.1.3 Pipeline Vaccines
8.1.3.1 CVnCoV
8.1.3.2 LUNAR-COV19
8.1.3.3 MRT5500
8.1.3.4 DS-5670
8.2 Non-COVID-19 mRNA Vaccines
8.2.1 Overview
8.2.2 Oncology
8.2.3 Infectious Diseases
8.2.4 Other Diseases
9.1 Overview
9.2 COVID-19 (by mRNA Type)
9.2.1 Nucleoside-Modified mRNA (modRNA)
9.2.2 Unmodified mRNA
9.2.3 Self-Amplifying mRNA
9.3 Non-COVID-19 (mRNA Type)
9.3.1 Nucleoside-Modified mRNA (modRNA)
9.3.2 Unmodified mRNA
9.3.3 Self-Amplifying mRNA
10.1 Overview
10.2 North America
10.2.1 North America mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines)
10.2.1.1 U.S.
10.2.1.2 Canada
10.2.2 North America mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines)
10.3 Europe
10.3.1 Europe mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines)
10.3.1.1 Germany
10.3.1.2 France
10.3.1.3 U.K.
10.3.1.4 Italy
10.3.1.5 Spain
10.3.1.6 Rest-of-Europe
10.3.2 Europe mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines)
10.4 Asia-Pacific
10.4.1 Asia-Pacific mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines)
10.4.1.1 China
10.4.1.2 Japan
10.4.1.3 South Korea
10.4.1.4 Australia
10.4.1.5 Rest-of-Asia-Pacific
10.4.2 Asia-Pacific mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines)
10.5 Latin America
10.5.1 Latin America mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines)
10.5.1.1 Brazil
10.5.1.2 Mexico
10.5.1.3 Rest-of-Latin America
10.5.2 Latin America mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines)
10.6 Rest-of-the-World
10.6.1 Rest-of-the-World mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines)
10.6.2 Rest-of-the-World mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines)
11.1 Overview
11.2 Arcturus Therapeutics Holdings Inc.
11.2.1 Company Overview
11.2.2 Role of Arcturus Therapeutics Holdings Inc. in the Global mRNA Vaccines and Therapeutics Market
11.2.3 Financials
11.2.4 Key Insights About Financial Health of the Company
11.2.5 SWOT Analysis
11.3 BioNTech SE
11.3.1 Company Overview
11.3.2 Role of BioNTech SE in the Global mRNA Vaccines and Therapeutics Market
11.3.3 Financials
11.3.4 Key Insights About Financial Health of the Company
11.3.5 SWOT Analysis
11.4 CureVac N.V.
11.4.1 Company Overview
11.4.2 Role of CureVac N.V. in the Global mRNA Vaccines and Therapeutics Market
11.4.3 Financials
11.4.4 Key Insights About Financial Health of the Company
11.4.5 SWOT Analysis
11.5 Daiichi Sankyo Company, Limited.
11.5.1 Company Overview
11.5.2 Role of Daiichi Sankyo Company, Limited. in the Global mRNA Vaccines and Therapeutics Market
11.5.3 Financials
11.5.4 Key Insights About Financial Health of the Company
11.5.5 SWOT Analysis
11.6 ethris GmbH
11.6.1 Company Overview
11.6.2 Role of ethris GmbH in Global mRNA Vaccines and Therapeutics Market
11.6.3 SWOT Analysis
11.7 GlaxoSmithKline plc
11.7.1 Company Overview
11.7.2 Role of GlaxoSmithKline plc in Global mRNA Vaccines and Therapeutics Market
11.7.3 Financials
11.7.4 Key Insights About Financial Health of the Company
11.7.5 SWOT Analysis
11.8 Gennova Biopharmaceuticals Ltd
11.8.1 Company Overview
11.8.2 Role of Gennova Biopharmaceuticals Ltd in Global mRNA Vaccines and Therapeutics Market
11.8.3 SWOT Analysis
11.9 Moderna, Inc.
11.9.1 Company Overview
11.9.2 Role of Moderna, Inc. in the Global mRNA Vaccines and Therapeutics Market
11.9.3 Financials
11.9.4 Key Insights About Financial Health of the Company
11.9.5 SWOT Analysis
11.10 Pantherna Therapeutics GmbH
11.10.1 Company Overview
11.10.2 Role of Pantherna Therapeutics GmbH in Global mRNA Vaccines and Therapeutics Market
11.10.3 SWOT Analysis
11.11 Providence Therapeutics
11.11.1 Company Overview
11.11.2 Role of Providence Therapeutics in Global mRNA Vaccines and Therapeutics Market
11.11.3 SWOT Analysis
11.12 Silence Therapeutics
11.12.1 Company Overview
11.12.2 Role of Silence Therapeutics in the Global mRNA Vaccines and Therapeutics Market
11.12.3 Financials
11.12.4 Key Insights About Financial Health of the Company
11.12.5 SWOT Analysis
11.13 Translate Bio
11.13.1 Company Overview
11.13.2 Role of Translate Bio in the Global mRNA Vaccines and Therapeutics Market
11.13.3 Financials
11.13.4 Key Insights About Financial Health of the Company
11.13.5 SWOT Analysis
11.14 VERSAMEB AG
11.14.1 Company Overview
11.14.2 Role of VERSAMEB AG in the Global mRNA Vaccines and Therapeutics Market
11.14.3 SWOT Analysis
11.15 Verve Therapeutics, Inc.
11.15.1 Company Overview
11.15.2 Role of Verve Therapeutics, Inc. in the Global mRNA Vaccines and Therapeutics Market
11.15.3 SWOT Analysis
Table 6.1: Likert Scale
Table 6.2: Impact Analysis of Market Drivers
Table 6.3: Impact Analysis of Market Restraints
Table 8.1: mRNA Vaccines and Therapeutics for Oncology (Pipeline)
Table 8.2: mRNA Vaccines and Therapeutics for Infectious Diseases (Pipeline)
Table 8.3: mRNA Vaccines and Therapeutics for Other Diseases (Pipeline)
Figure 1: Cumulative COVID-19 Cases (as of April 18, 2021)
Figure 2: Impact Analysis of Market Drivers and Market Challenges on Global mRNA Vaccines and Therapeutics Market
Figure 3: Global mRNA Vaccines and Therapeutics Market for COVID-19 Vaccines (Commercialized), Q4 2020 Vs. Q4 2025
Figure 4: Global mRNA Vaccines and Therapeutics Market for COVID-19 Vaccines (Pipeline), Q2 2021 Vs. Q4 2025
Figure 5: Global mRNA Vaccines and Therapeutics Market for Non-COVID-19 Vaccines (Pipeline), 2025 Vs. 2031
Figure 6: Global mRNA Vaccines and Therapeutics Market (by mRNA Type)
Figure 7: Global mRNA Vaccines and Therapeutics Market for COVID-19 Vaccines (by Region), 2020-2025
Figure 8: Global mRNA Vaccines and Therapeutics Market for Non-COVID-19 Vaccines (by Region), 2025-2031
Figure 2.1: Global mRNA Vaccines and Therapeutics, Market Segmentation
Figure 3.1: Global mRNA Vaccines and Therapeutics Market Research Methodology
Figure 3.2: Primary Research Methodology
Figure 3.3: Bottom-Up Approach (Segment-Wise Analysis)
Figure 3.4: Top-Down Approach (Segment-Wise Analysis)
Figure 4.1: Global Revenue for COVID-19 mRNA Vaccines, 2020-2025
Figure 4.2: Global mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines), 2025-2031
Figure 5.1: Steps for Obtaining Marketing Authorization
Figure 5.2: Vaccine Development Stages
Figure 5.3: Steps for Obtaining EMA Approval for Vaccines
Figure 6.1: Cumulative COVID-19 Cases (as of April 18, 2021)
Figure 6.2: Logistical Challenge Pertaining to mRNA-Based COVID-19 Vaccines
Figure 7.1: Share of Key Developments and Strategies, January 2018-March 2021
Figure 7.2: Share of Synergistic Activities (by Company), January 2018-March 2021
Figure 7.3: Share of Product Approvals and Commercialization (by Company), January 2018-March 2021
Figure 7.4: Share of Funding (by Company), January 2018-March 2021
Figure 7.5: Market Share Analysis for Global mRNA Vaccines and Therapeutics Market, 2020 and 2021
Figure 8.1: Global Revenue for COVID-19 mRNA Vaccines, $Million, 2020-2025
Figure 8.2: Global Revenue for COVID-19 mRNA Vaccines (Commercialized), $Million, Q4 2020-Q4 2025
Figure 8.3: Global Revenue for COVID-19 mRNA Vaccines (mRNA-1273), $Million, Q4 2020-Q4 2025
Figure 8.4: Global Revenue for COVID-19 mRNA Vaccines (BNT162b2), $Million, Q4 2020-Q4 2025
Figure 8.5: Expected Revenue from mRNA-Based COVID-19 Vaccines (Pipeline), $Million, Q4 2020-Q4 2025
Figure 8.6: Global Estimated Revenue for CVnCoV, $Million, Q2 2021- Q4 2025
Figure 8.7: Global Estimated Revenue for LUNAR-COV19, $Million, Q3 2022-Q4 2025
Figure 8.8: Global Estimated Revenue for MRT5500, $Million, Q4 2022-Q4 2025
Figure 8.9: Global Estimated Revenue for DS-5670, $Million, Q4 2022-Q4 2025
Figure 8.10: Global mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines)
Figure 8.11: Global mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines), 2025-2031
Figure 8.12: Global mRNA Vaccines and Therapeutics Market (Oncology), $Million, 2025-2031
Figure 8.13: Global mRNA Vaccines and Therapeutics Market (Infectious Diseases), $Million, 2025-2031
Figure 8.14: Global mRNA Vaccines and Therapeutics Market (Other Diseases), $Million, 2025-2031
Figure 9.1: Global mRNA Vaccines and Therapeutics Market (by mRNA Type)
Figure 9.2: Global mRNA Vaccines and Therapeutics Market for COVID-19 (Nucleoside-Modified mRNA), $Million, Q42020-Q42025
Figure 9.3: Global mRNA Vaccines and Therapeutics Market for COVID-19 (Unmodified mRNA), $Million, Q2 2021-Q4 2025
Figure 9.4: Global mRNA Vaccines and Therapeutics Market for COVID-19 (Self-Amplifying mRNA), $Million, Q3 2022-Q4 2025
Figure 9.5: Global mRNA Vaccines and Therapeutics Market for Non-COVID-19 (Nucleoside-Modified mRNA), $Million, 2025-2031
Figure 9.6: Global mRNA Vaccines and Therapeutics Market for Non-COVID-19 (Unmodified mRNA), $Million, 2025-2031
Figure 9.7: Global mRNA Vaccines and Therapeutics Market for Non-COVID-19 (Self-Amplifying mRNA), $Million, 2029-2031
Figure 10.1: Global mRNA Vaccines and Therapeutics Market for COVID-19 Vaccines (by Region), 2020 and 2025
Figure 10.2: Global mRNA Vaccines and Therapeutics Market for Non-COVID-19 Vaccines (by Region), 2025 and2031
Figure 10.3: North America: Market Dynamics
Figure 10.4: North America mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020 vs. Q4 2025
Figure 10.5: North America mRNA Vaccines and Therapeutics Market (COVID-19 Vaccines), (by Country), $Million, Q4 2020 vs. Q4 2025
Figure 10.6: U.S. mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.7: Canada mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.8: North America mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines), $Million, 2025-2031
Figure 10.9: Europe: Market Dynamics
Figure 10.10: Europe mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.11: Europe mRNA Vaccines and Therapeutics Market (COVID-19 Vaccines), (by Country), $Million, Q4 2020 vs. Q4 2025
Figure 10.12: Germany mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.13: France mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.14: U.K. mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.15: Italy mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.16: Spain mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.17: Rest-of-Europe mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q1 2020-Q4 2025
Figure 10.18: Europe mRNA Vaccines and Therapeutics (by Non-COVID-19 Vaccines), $Million, 2025-2031
Figure 10.19: Asia-Pacific: Market Dynamics
Figure 10.20: Asia-Pacific mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.21: Asia-Pacific mRNA Vaccines and Therapeutics Market (COVID-19 Vaccines), (by Country), $Million, Q1 2021 vs. Q4 2025
Figure 10.22: China mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q2 2021-Q4 2025
Figure 10.23: Japan mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q1 2021-Q4 2025
Figure 10.24: South Korea mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q2 2021-Q4 2025
Figure 10.25: Australia mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q1 2021-Q4 2025
Figure 10.26: Rest-of-Asia-Pacific mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q1 2021-Q4 2025
Figure 10.27: Asia-Pacific mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines), $Million, 2026-2031
Figure 10.28: Latin America: Market Dynamics
Figure 10.29: Latin America mRNA Vaccines and Therapeutics Market (COVID-19 Vaccines), (by Country), $Million, Q1 2021 vs. Q4 2025
Figure 10.30: Latin America mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.31: Brazil mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q1 2021-Q4 2025
Figure 10.32: Mexico mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q1 2021-Q4 2025
Figure 10.33: Rest-of-Latin America mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q1 2021-Q4 2025
Figure 10.34: Latin America mRNA Vaccines and Therapeutics Market (by Non-COVID-19 Vaccines), $Million, 2026-2031
Figure 10.35: Rest-of-the-World mRNA Vaccines and Therapeutics Market (by COVID-19 Vaccines), $Million, Q4 2020-Q4 2025
Figure 10.36: Rest-of-the-World mRNA Vaccines and Therapeutics (by Non-COVID-19 Vaccines), $Million, 2026-2031
Figure 11.1: Total Number of Companies Profiled
Figure 11.2: Arcturus Therapeutics Holdings Inc.: Pipeline Portfolio
Figure 11.3: Arcturus Therapeutics Holdings Inc.: Overall Financials, 2018-2020
Figure 11.4: Arcturus Therapeutics Holdings Inc.: R&D Expenditure, 2018-2020
Figure 11.5: Arcturus Therapeutics Holdings Inc.: SWOT Analysis
Figure 11.6: BioNTech SE: Product Portfolio
Figure 11.7: BioNTech SE: Pipeline Portfolio
Figure 11.8: BioNTech SE: Overall Financials, 2018-2020
Figure 11.9: BioNTech SE: Revenue (by Segment), 2018-2020
Figure 11.10: BionNTech SE: R&D Expenditure, 2018-2020
Figure 11.11: BioNTech SE: SWOT Analysis
Figure 11.12: CureVac N.V.: Pipeline Portfolio
Figure 11.13: CureVac N.V.: Overall Financials, 2018-2019
Figure 11.14: CureVac N.V.: Revenue (by Region), 2018-2019
Figure 11.15: CureVac N.V.: R&D Expenditure, 2018-2019
Figure 11.16: CureVac N.V.: SWOT Analysis
Figure 11.17: Daiichi Sankyo Company, Limited: Pipeline Portfolio
Figure 11.18: Daiichi Sankyo Company, Limited: Overall Financials, 2017-2019
Figure 11.19: Daiichi Sankyo Company, Limited.: Revenue (by Segment), 2017-2019
Figure 11.20: Daiichi Sankyo Company, Limited.: Revenue (by Region), 2017-2019
Figure 11.21: Daiichi Sankyo Company, Limited: R&D Expenditure, 2017-2019
Figure 11.22: Daiichi Sankyo Company, Limited.: SWOT Analysis
Figure 11.23: ethris GmbH: Pipeline Portfolio
Figure 11.24: ethris GmbH: SWOT Analysis
Figure 11.25: GlaxoSmithKline plc: Pipeline Portfolio
Figure 11.26: GlaxoSmithKline plc: Overall Financials, 2018-2020
Figure 11.27: GlaxoSmithKline plc: Revenue (by Segment), 2018-2020
Figure 11.28: GlaxoSmithKline plc: Revenue (by Region), 2018-2020
Figure 11.29: GlaxoSmithKline plc: R&D Expenditure, 2018-2020
Figure 11.30: GlaxoSmithKline plc: SWOT Analysis
Figure 11.31: Gennova Biopharmaceuticals Ltd: Pipeline Portfolio
Figure 11.32: Gennova Biopharmaceuticals Ltd: SWOT Analysis
Figure 11.33: Moderna, Inc.: Product Portfolio
Figure 11.34: Moderna, Inc: Pipeline Portfolio
Figure 11.35: Moderna, Inc.: Overall Financials, 2018-2020
Figure 11.36: Moderna, Inc.: Revenue (by Segment), 2018-2020
Figure 11.37: Moderna, Inc.: Revenue (by Region), 2020
Figure 11.38: Moderna, Inc.: R&D Expenditure, 2018-2020
Figure 11.39: Moderna, Inc.: SWOT Analysis
Figure 11.40: Pantherna Therapeutics GmbH: Pipeline Portfolio
Figure 11.41: Pantherna Therapeutics GmbH: SWOT Analysis
Figure 11.42: Providence Therapeutics: Pipeline Portfolio
Figure 11.43: Providence Therapeutics: SWOT Analysis
Figure 11.44: Silence Therapeutics: Pipeline Portfolio
Figure 11.45: Silence Therapeutics: Overall Financials, 2019-2020
Figure 11.46: Silence Therapeutics: Revenue (by Segment), 2019-2020
Figure 11.47: Silence Therapeutics: Revenue (by Region), 2019-2020
Figure 11.48: Silence Therapeutics: R&D Expenditure, 2019-2020
Figure 11.49: Silence Therapeutics: SWOT Analysis
Figure 11.50: Translate Bio: Pipeline Portfolio
Figure 11.51: Translate Bio: Overall Financials, 2018-2020
Figure 11.52: Translate Bio: R&D Expenditure, 2018-2020
Figure 11.53: Translate Bio: SWOT Analysis
Figure 11.54: VERSAMEB AG: Pipeline Portfolio
Figure 11.55: VERSAMEB AG: SWOT Analysis
Figure 11.56: Verve Therapeutics, Inc.: Pipeline Portfolio
Figure 11.57: Verve Therapeutics, Inc.: SWOT Analysis
Report Description
|
Market Report Coverage – mRNA Vaccines and Therapeutics |
|||||
|
|
COVID-19 Vaccines |
Non-COVID-19 Vaccines |
|
COVID-19 Vaccines |
Non-COVID-19 Vaccines |
|
Base Year |
2020 |
2025 |
Market Size in 2021-2026 |
$51.65 Billion |
$0.05 Billion |
|
Forecast Period |
2021-2025 |
2026 - 2031 |
Value Projection and Estimation by 2025-2031 |
$28.92 Billion |
$1.69 Billion |
|
CAGR During Forecast Period |
-13.50% |
95.49% |
Number of Tables |
06 |
|
|
Number of Pages |
207 |
Number of Figures |
139 |
||
|
Research Hours |
240 Hours |
||||
|
Market Segmentation |
• Application – COVID-19 mRNA Vaccines and Non-COVID-19 mRNA Vaccines • mRNA Type – Nucleoside-Modified mRNA, Unmodified mRNA, and Self-Amplifying mRNA |
||||
|
Regional Segmentation |
• North America – U.S., and Canada • Europe – Germany, France, U.K., Italy, Spain, and Rest-of-Europe • Asia-Pacific – China, Japan, South Korea, Australia, and Rest-of-Asia-Pacific • Latin America – Brazil, Mexico, and Rest-of-Latin America • Rest-of-the-World |
||||
|
Growth Drivers |
• Growing Cases of COVID-19 Globally • Favorable Regulatory Environment • Significant External Funding for the Development of mRNA-Based COVID-19 Vaccines |
||||
|
Market Challenges |
• Logistical Challenges Associated with mRNA-Based COVID-19 Vaccines • High Price Associated with mRNA-Based COVID-19 Vaccines |
||||
|
Market Opportunities |
• Growing Emphasis on Research and Development for Non-COVID-19 Applications |
||||
|
Key Companies Profiled |
Arcturus Therapeutics Holdings Inc., BioNTech SE, CureVac N.V., Daiichi Sankyo Company, Limited., ethris GmbH, GlaxoSmithKline plc, Gennova Biopharmaceuticals Ltd, Moderna, Inc., Pantherna Therapeutics GmbH, Providence Therapeutics, Silence Therapeutics, Translate Bio, VERSAMEB AG, and Verve Therapeutics, Inc. |
||||
Key Questions Answered in this Report:
- What are the major market drivers, challenges, and opportunities in the global mRNA vaccines and mRNA therapeutics market?
- What are the key development strategies being implemented by the major players in order to sustain in the competitive market?
- What are the key regulatory implications in developed and developing regions for the global mRNA vaccines and mRNA therapeutics market?
- How is each segment of the market expected to grow during the forecast period, 2021-2031?
- Who are the leading players with significant offerings to the global mRNA vaccines and mRNA therapeutics market, and what is the expected market dominance for each of these leading players?
- What are the adoption trends for mRNA vaccines and mRNA therapeutics in established economies and emerging economies across the world?
- Which companies are anticipated to be highly disruptive in the future, and why?
- What are the current clinical unmet needs that are being faced in the global mRNA vaccines and mRNA therapeutics market?
- What are the dynamics of various application areas and countries, which are impacting the global mRNA vaccines and mRNA therapeutics market?
- What are the new market opportunities that are influencing the growth of the global mRNA vaccines and mRNA therapeutics market?
- How is each segment of the market expected to grow during the forecast period from 2021 to 2031? Following are the segment types:
- Application
- mRNA Type
- Region
- What are the unmet needs currently existing in the global mRNA vaccines and mRNA therapeutics market?
Market Overview
BIS healthcare experts have found that the global market for mRNA vaccines and therapeutics to be one of the most rapidly evolving and dynamic markets, and the global mRNA vaccines market for COVID-19 is predicted to decline at a CAGR of 13.50% over the forecast period of 2021-2025. The global mRNA vaccines market is driven by certain factors, which include growing cases of COVID-19 globally, a favorable regulatory environment, and significant external funding for the development of mRNA-based COVID-19 vaccines. Moreover, the global mRNA therapeutics market for non-COVID-19 applications is predicted to grow at a CAGR of 95.49% over the forecast period of 2026-2031. The global mRNA therapeutics market is majorly driven by the increasing focus on research in the field of mRNA for deciphering insights regarding its clinical utility, growing fund infusions by the government-funded initiatives, and growing product pipeline, among others.
Figure: Global mRNA Vaccines Market for COVID-19 Vaccines Snapshot
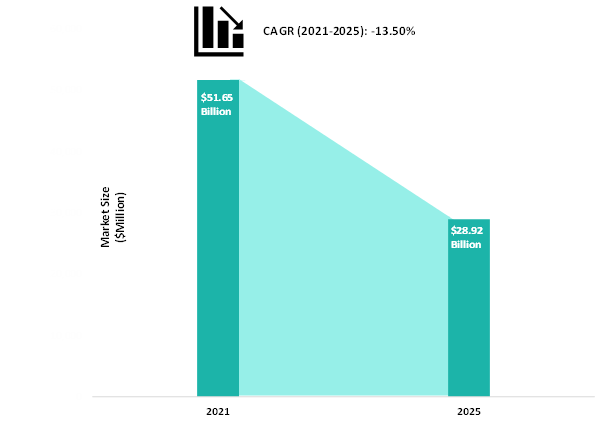
The global mRNA vaccines market for COVID-19 is favored by the developments in the field of mRNA-based COVID-19 vaccines due to the growing cases of COVID-19 globally, favourable regulatory environment, and significant external funding for the development of mRNA-based COVID-19 vaccines. Additionally, the emergence of the new SARS-CoV-2 virus in different countries, coupled with the efficacy of mRNA vaccines against new strains, are some of the critical factors which are expected to bolster the growth of the global mRNA vaccines market, particularly during, Q4 2020- Q4 2022. Furthermore, pharmaceutical companies are focusing on the development of new mRNA-based vaccine candidates for COVID-19 to support the healthcare authorities and the government to establish robust vaccination strategies. However, due to the expected transition of the ongoing COVID-19 pandemic into an endemic by the end of 2023, the market is anticipated to drastically decline by the end of 2025.
Figure: Global mRNA Therapeutics Market for Non-COVID-19 Vaccines Snapshot
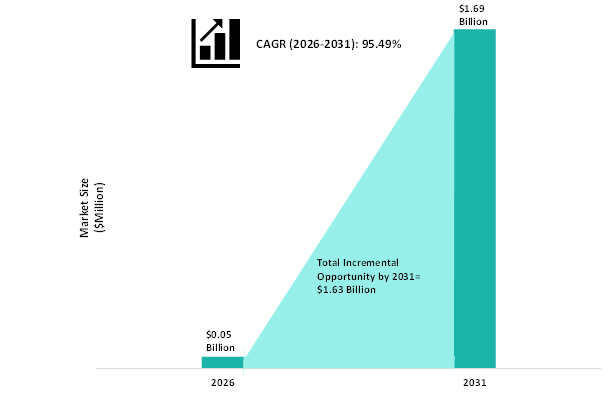
The global mRNA therapeutics market for non-COVID-19 applications is favored by the clinical developments which have been taking place to development of various therapies for non-COVID-19 applications, particularly for infectious disease, oncology, and other diseases. It should be noted here that the global mRNA therapeutics market consists of vaccines which are being developed for non-COVID-19 applications. The emerging and legacy pharmaceutical companies are massively investing in the development of these treatment modalities to further expand their pipeline. The successful launch of these candidates after obtaining regulatory approvals from the regulatory authorities is anticipated to increase the competitiveness of the global mRNA therapeutics market in the forthcoming years.
Within the research report, the market is segmented on the basis of applications, mRNA type, and region. Each of these segments covers the snapshot of the market over the projected years, the inclination of the market revenue, underlying patterns, and trends by using analytics on the primary and secondary data obtained.
Competitive Landscape
The exponential rise in the number of COVID-19 cases followed by the growing emphasis of the government for establishing robust vaccination strategies has created a buzz among the pharmaceutical companies to further invest in the development of mRNA-based vaccines for COVID-19. Additionally, due to the growing prominence of mRNA vaccines, pharmaceutical companies are also significantly investing to expand their respective pipeline for non-COVID-19 applications such as infectious diseases, oncology, and other diseases. Due to the presence of a diverse portfolio and intense market penetration, BioNTech SE has been a pioneer in the mRNA vaccines and therapeutics ecosystem.
On the basis of region, North America holds the largest share of the global mRNA vaccines market for COVID-19 due to significantly growing fund infusions for mRNA vaccine development, high rise in the pipeline products, presence of established and emerging pharmaceutical companies, presence of ultracold manufacturing and supply chains, and increasing awareness about mRNA-based COVID-19 vaccines, among others. Apart from this, the Asia-Pacific region is anticipated to grow at the fastest rate due to the exponentially rising cases of COVID-19 in the emerging countries, presence of a high population base in the region, and increasing demand for mRNA-based vaccines, among others.
Moreover, North America holds the largest share of the global mRNA therapeutics market for non-COVID-19 applications due to the growing need to develop transformative therapeutics, increasing fund infusions, a favorable regulatory environment, and massively growing attention to establish precision medicine, among others.
Global mRNA Vaccines and Therapeutics Market
Focus on Application, mRNA Type, Country Data (13 Countries), and Competitive Landscape - Analysis and Forecast, 2021-2031
Frequently Asked Questions
The global mRNA vaccines and therapeutics market is expected to grow in negative in (-13.50%) during 2021-2025 for the Covid Vaccines. In case of Non-Covid Vaccine the market is expected to grow at a CAGR of 95.49% during 2026-2031.
Challenges that are hindering the growth of the market include logistical challenges associated with mRNA-based covid-19 vaccines and high price associated with mRNA-based covid-19 vaccines.
Factors driving the growth of the market include growing cases of covid-19 globally, favorable regulatory environment, and significant external funding for the development of mRNA-based covid-19 vaccines.
The mRNA vaccines are used in both in COVID-19 and non-COVID-19 vaccines.
Arcturus Therapeutics Holdings Inc., BioNTech SE, CureVac N.V., Daiichi Sankyo Company, Limited., ethris GmbH, GlaxoSmithKline plc, Gennova Biopharmaceuticals Ltd, Moderna, Inc., Pantherna Therapeutics GmbH, Providence Therapeutics, Silence Therapeutics, Translate Bio, VERSAMEB AG, and Verve Therapeutics, Inc.

