A quick peek into the report
Table of Contents
1.1 Product Definition
1.2 Inclusion and Exclusion Criteria
1.3 Market Scope
1.3.1 Scope of the Study
1.3.2 Key Questions Answered in the Report
1.4 Research Methodology
1.4.1 Global Multicancer Screening Market: Research Methodology
1.4.2 Data Sources
1.4.3 Market Estimation Model
1.4.4 Criteria for Company Profiling
2.1 Market Overview
2.2 Multicancer Screening Workflow Analysis
2.3 Market Footprint and Growth Potential
2.4 Future Potential
2.5 COVID-19 Impact on the Market
3.1 Patent Analysis
3.1.1 Patent Filing Trend
3.1.2 Patent Analysis (by Country)
3.1.3 Patent Analysis (by Region)
3.2 Reimbursement Scenario
3.3 Supply Chain Analysis
3.4 Pricing Analysis (by Test Type)
4.1 Overview
4.2 Impact Analysis
4.3 Market Drivers
4.3.1 Early Detection of Cancer Leads to Higher Survival Chances and Better Treatment
4.3.2 Increasing Popularity of Liquid Biopsy Leading to Easier Screening
4.3.3 Reduced Treatment Cost Due to Earlier Cancer Detection
4.4 Market Challenges
4.4.1 Lack of Awareness about the Multicancer Screening Tests
4.4.2 Too Costly to be Used as Preventive Care
4.5 Market Opportunities
4.5.1 Robust Pipeline of Multicancer Screening Tests Poised to Revolutionize Cancer Diagnosis
4.5.2 New Biomarkers being Discovered Regularly
5.1 Key Strategies and Developments
5.1.1 Mergers and Acquisitions
5.1.2 Synergistic Activities
5.1.3 Product Launches and Upgradations
5.1.4 Funding and Expansion
5.2 Market Share Analysis (2022)
5.3 Growth-Share Analysis (2022)
5.3.1 Growth-Share Analysis (by Technology)
6.1 Overview
6.2 Laboratory Developed Tests (LDTs)
6.3 In-Vitro Diagnostics (IVDs)
7.1 Overview
7.2 Next-Generation Sequencing (NGS)
7.3 Polymerase Chain Reaction (PCR)
7.4 Immunohistochemistry (IHC)
7.5 Fluorescence In-Situ Hybridization (FISH)
7.6 Other Technologies
8.1 Overview
8.2 Tissue
8.3 Blood
8.4 Saliva and Buccal Swab
8.5 Others
9.1 Overview
9.2 DNA
9.3 RNA
9.4 Proteins
10.1 Overview
10.2 Breast and Gynecologic
10.3 Gastrointestinal
10.4 Endocrine
10.5 Genitourinary
10.6 Skin
10.7 Brain/Nervous System
10.8 Sarcoma
10.9 Hematological Malignancies
10.10 Lung
10.11 Head and Neck
10.12 Other Cancer Types
11.1 Overview
11.2 Clinical
11.3 Research
12.1 Overview
12.2 Hospitals
12.3 Diagnostic and Clinical Laboratories
12.4 Academic Research Institutions
12.5 Other End Users
13.1 Overview
13.2 North America
13.2.1 U.S.
13.2.2 Canada
13.3 Europe
13.3.1 Germany
13.3.2 France
13.3.3 U.K.
13.3.4 Italy
13.3.5 Spain
13.3.6 Rest-of-Europe
13.4 Asia-Pacific
13.4.1 China
13.4.2 Japan
13.4.3 India
13.4.4 Australia
13.4.5 South Korea
13.4.6 Rest-of-Asia-Pacific
13.5 Latin America
13.5.1 Brazil
13.5.2 Mexico
13.5.3 Rest-of-Latin America
13.6 Rest-of-the-World
14.1 Overview
14.2 Market Value Chain of Multicancer Screening Market
14.3 Multicancer Screening Ecosystem Active Players
14.4 Agilent Technologies, Inc.
14.4.1 Company Overview
14.4.2 Role of Agilent Technologies, Inc. in the Global Multicancer Screening Market
14.4.3 Key Competitors of the Company
14.4.4 Financials
14.4.5 Key Insights about the Financial Health of the Company
14.4.6 Analyst Perspective
14.5 Atara Biotherapeutics, Inc.
14.5.1 Company Overview
14.5.2 Role of Atara Biotherapeutics, Inc. in the Global Multicancer Screening Market
14.5.3 Key Competitors of the Company
14.5.4 Financials
14.5.5 Key Insights about the Financial Health of the Company
14.5.6 Analyst Perspective
14.6 Burning Rock DX
14.6.1 Company Overview
14.6.2 Role of Burning Rock DX in the Global Multicancer Screening Market
14.6.3 Key Competitors of the Company
14.6.4 Business Strategies
14.6.5 Analyst Perspective
14.7 Danaher Corporation (Integrated DNA Technologies, Inc.)
14.7.1 Company Overview
14.7.2 Role of Danaher Corporation (Integrated DNA Technologies, Inc.) in the Global Multicancer Screening Market
14.7.3 Key Competitors of the Company
14.7.4 Financials
14.7.5 Key Insights about the Financial Health of the Company
14.7.6 Analyst Perspective
14.8 Exact Sciences Corporation
14.8.1 Company Overview
14.8.2 Role of Exact Sciences Corporation in the Global Multicancer Screening Market
14.8.3 Key Competitors of the Company
14.8.4 Financials
14.8.5 Key Insights about the Financial Health of the Company
14.8.6 Corporate Strategies
14.8.7 Business Strategies
14.8.8 Analyst Perspective
14.9 F. Hoffmann-La Roche Ltd
14.9.1 Company Overview
14.9.2 Role of F. Hoffmann-La Roche Ltd in the Global Multicancer Screening Market
14.9.3 Key Competitors of the Company
14.9.4 Financials
14.9.5 Key Insights about the Financial Health of the Company
14.9.6 Corporate Strategies
14.9.7 Business Strategies
14.9.8 Analyst Perspective
14.10 Fulgent Genetics.
14.10.1 Company Overview
14.10.2 Role of Fulgent Genetics. in the Global Multicancer Screening Market
14.10.3 Key Competitors of the Company
14.10.4 Financials
14.10.5 Key Insights about the Financial Health of the Company
14.10.6 Analyst Perspective
14.11 Genecast Biotechnology Co., Ltd
14.11.1 Company Overview
14.11.2 Role of Genecast Biotechnology Co., Ltd in the Global Multicancer Screening Market
14.11.3 Key Competitors of the Company
14.11.4 Analyst Perspective
14.12 Guardant Health, Inc.
14.12.1 Company Overview
14.12.2 Role of Guardant Health, Inc. in the Global Multicancer Screening Market
14.12.3 Key Competitors of the Company
14.12.4 Financials
14.12.5 Key Insights about the Financial Health of the Company
14.12.6 Business Strategies
14.12.7 Analyst Perspective
14.13 Illumina, Inc. (GRAIL, LLC.)
14.13.1 Company Overview
14.13.2 Role of Illumina, Inc. (GRAIL, LLC.) in the Global Multicancer Screening Market
14.13.3 Key Competitors of the Company
14.13.4 Financials
14.13.5 Key Insights about the Financial Health of the Company
14.13.6 Corporate Strategies
14.13.7 Analyst Perspective
14.14 Konica Minolta, Inc. (Ambry Genetics.)
14.14.1 Company Overview
14.14.2 Role of Konica Minolta, Inc. (Ambry Genetics.) in the Global Multicancer Screening Market
14.14.3 Key Competitors of the Company
14.14.4 Financials
14.14.5 Key Insights about the Financial Health of the Company
14.14.6 Analyst Perspective
14.15 Laboratory Corporation of America Holdings.
14.15.1 Company Overview
14.15.2 Role of Laboratory Corporation of America Holdings. in Global Multicancer Screening Market
14.15.3 Key Competitors of the Company
14.15.4 Financials
14.15.5 Corporate Strategies
14.15.6 Business Strategies
14.15.7 Analyst’s Perspective
14.16 Myriad Genetics, Inc.
14.16.1 Company Overview
14.16.2 Role of Myriad Genetics, Inc. in the Global Multicancer Screening Market
14.16.3 Key Competitors of the Company
14.16.4 Financials
14.16.5 Key Insights about the Financial Health of the Company
14.16.6 Business Strategies
14.16.7 Analyst Perspective
14.17 Tempus
14.17.1 Company Overview
14.17.2 Role of Tempus in the Global Multicancer Screening Market
14.17.3 Key Competitors of the Company
14.17.4 Analyst Perspective
14.18 Thermo Fisher Scientific Inc.
14.18.1 Company Overview
14.18.2 Role of Thermo Fisher Scientific Inc. in the Global Multicancer Screening Market
14.18.3 Key Competitors of the Company
14.18.4 Financials
14.18.5 Key Insights about the Financial Health of the Company
14.18.6 Analyst Perspective
14.19 Emerging Companies
14.19.1 Freenome Holdings, Inc.
14.19.2 Invitae Corporation.
14.19.3 Caris Life Sciences.
14.19.4 Oncocyte Corporation
Table 1: Impact Analysis of Market Drivers, Challenges, and Opportunities on the Global Multicancer Screening Market
Table 2: Technological Trends in Global Multicancer Screening Market
Table 3: Average Prices of Multicancer Screening Kits Calculation
Table 4: Likert Scale
Table 5: Impact Analysis of Market Drivers
Table 6: Impact Analysis of Market Challenges
Table 7: Five-Year Survival Rates for Different Cancers at Different Stages
Table 8: Top Multicancer Screening Tests and Their Costs ($)
Table 9: Global Multicancer Screening Market, Pipeline Tests
Table 10: Cancers and Their Most Common Biomarker Tests
Table 11: Laboratory Developed Tests (LDTs) vs. In-Vitro Diagnostics (IVDs)
Table 12: DNA vs. RNA vs. Protein Products Heat Map
Table 13: NGS Approach for B and T-Acute Lymphoblastic Leukemia (ALL)
Figure 1: Global Multicancer Screening Market, $Billion, 2022 and 2032
Figure 2: Share of Global Multicancer Screening Market (by Test Type), $Billion, 2021 and 2032
Figure 3: Share of Global Multicancer Screening Market (by Sample), $Billion, 2021 and 2032
Figure 4: Share of Global Multicancer Screening Market (by Cancer Type), $Billion, 2021 and 2032
Figure 5: Share of Global Multicancer Screening Market (by Region), 2022
Figure 6: Global Multicancer Screening Market Segmentation
Figure 7: Global Multicancer Screening Market: Research Methodology
Figure 8: Primary Research Methodology
Figure 9: Bottom-Up Approach (Segment-Wise Analysis)
Figure 10: Top-Down Approach (Segment-Wise Analysis)
Figure 11: Multicancer Screening Workflow, End User Perspective
Figure 12: Multicancer Screening Workflow, Laboratory Technician Perspective
Figure 13: Global Multicancer Screening Market, $Billion, 2021-2032
Figure 14: COVID-19 Impact
Figure 15: Global Multicancer Screening Market, Patent Analysis (by Year), January 2020-December 2022
Figure 16: Global Multicancer Screening Market, Patent Analysis (by Country), January 2020-December 2022
Figure 17: Global Multicancer Screening Market, Patent Analysis (by Region), January 2020-December 2022
Figure 18: Supply Chain Analysis of the Global Multicancer Screening Market
Figure 19: Global Multicancer Screening Market Dynamics
Figure 20: Prevalence vs. Age-Standardized Death Rate of Cancer Globally, 2010-2019 (%)
Figure 21: Liquid Biopsy vs. Tissue Biopsy
Figure 22: Liquid Biopsy Clinical Trials (by Cancer Types), Share (%), 2023
Figure 23: Out-of-Pocket Costs for Complete Treatment by Cancer Type and Stage Among Insured Patient, ($)
Figure 24: Lead-Time Bias in Cancer Screening
Figure 25: Screened vs. Non-Screened Cancer Patients per 1,000,000 Incidence Rate (50-79 Years)
Figure 26: Multicancer Early Detection Tests + Usual Care Screening Costs vs. only Usual Care Screening Costs
Figure 27: Share of Key Developments and Strategies, January 2020-January 2023
Figure 28: Synergistic Activities Share (by Company), January 2020- January 2023
Figure 29: Product Launches and Upgradations (by Company), January 2020-January 2023
Figure 30: Market Share Analysis for Global Multicancer Screening Market, $Billion, 2022
Figure 31: Growth-Share Analysis of the Global Multicancer Screening Market (by Technology), 2022
Figure 32: Global Multicancer Screening Market (by Test Type)
Figure 33: Global Multicancer Screening Market (by Test Type), $Billion, 2021 and 2032
Figure 34: Global Multicancer Screening Market (Laboratory Developed Tests), $Billion, 2021-2032
Figure 35: Global Multicancer Screening Market (In-Vitro Diagnostics), $Billion, 2021-2032
Figure 36: Global Multicancer Screening Market (by Technology)
Figure 37: Global Multicancer Screening Market (by Technology), $Billion, 2021 and 2032
Figure 38: Global Multicancer Screening Market (Next-Generation Sequencing), $Billion, 2021-2032
Figure 39: Global Multicancer Screening Market (Polymerase Chain Reaction), $Billion, 2021-2032
Figure 40: Global Multicancer Screening Market (Immunohistochemistry), $Billion, 2021-2032
Figure 41: Global Multicancer Screening Market (Fluorescence In-Situ Hybridization), $Billion, 2021-2032
Figure 42: Global Multicancer Screening Market (Other Technologies), $Billion, 2021-2032
Figure 43: Liquid Biopsy Development Timeline (1948-2022)
Figure 44: Global Multicancer Screening Market (by Sample)
Figure 45: Global Multicancer Screening Market (by Sample), $Billion, 2021 and 2032
Figure 46: Global Multicancer Screening Market (Tissue), $Billion, 2021-2032
Figure 47: Global Multicancer Screening Market (Blood), $Billion, 2021-2032
Figure 48: Global Multicancer Screening Market (Saliva and Buccal Swab), $Billion, 2021-2032
Figure 49: Global Multicancer Screening Market (Others), $Billion, 2021-2032
Figure 50: Global Multicancer Screening Market (by Method)
Figure 51: Global Multicancer Screening Market (by Method), $Billion, 2021 and 2032
Figure 52: Global Multicancer Screening Market (DNA), $Billion, 2021-2032
Figure 53: Global Multicancer Screening Market (RNA), $Billion, 2021-2032
Figure 54: Global Multicancer Screening Market (Proteins), $Billion, 2021-2032
Figure 55: Global Multicancer Screening Market (by Cancer Type)
Figure 56: Global Multicancer Screening Market (by Cancer Type), $Billion, 2021 and 2032
Figure 57: Types of Breast and Gynecologic Cancers
Figure 58: Global Multicancer Screening Market (Breast and Gynecologic Cancer), $Billion, 2021-2032
Figure 59: Types of Gastrointestinal Cancers
Figure 60: Global Multicancer Screening Market (Gastrointestinal), $Billion, 2021-2032
Figure 61: Types of Endocrine Cancers
Figure 62: Global Multicancer Screening Market (Endocrine), $Million, 2021-2032
Figure 63: Types of Genitourinary Cancers
Figure 64: Global Multicancer Screening Market (Genitourinary), $Million, 2021-2032
Figure 65: Types of Skin Cancers
Figure 66: Global Multicancer Screening Market (Skin), $Million, 2021-2032
Figure 67: Global Multicancer Screening Market (Brain/Nervous System), $Million, 2021-2032
Figure 68: Global Multicancer Screening Market (Sarcoma), $Million, 2021-2032
Figure 69: Global Multicancer Screening Market (Hematological Malignancies), $Million, 2021-2032
Figure 70: Global Multicancer Screening Market (Lung), $Million, 2021-2032
Figure 71: Global Multicancer Screening Market (Head and Neck), $Million, 2021-2032
Figure 72: Global Multicancer Screening Market (Other Cancer Types), $Million, 2021-2032
Figure 73: Global Multicancer Screening Market (by Application)
Figure 74: Global Multicancer Screening Market (by Application), $Billion, 2021 and 2032
Figure 75: Global Multicancer Screening Market (Clinical), $Billion, 2021-2032
Figure 76: Global Multicancer Screening Market (Research), $Billion, 2021-2032
Figure 77: Global Multicancer Screening Market (by End User)
Figure 78: Global Multicancer Screening Market (by End User), $Billion, 2021 and 2032
Figure 79: Global Multicancer Screening Market (Hospitals), $Billion, 2021-2032
Figure 80: Global Multicancer Screening Market (Diagnostic and Clinical Laboratories), $Billion, 2021-2032
Figure 81: Global Multicancer Screening Market (Academic Research Institutions), $Billion, 2021-2032
Figure 82: Global Multicancer Screening Market (Other End Users), $Billion, 2021-2032
Figure 83: Global Multicancer Screening Market Snapshot (by Region), $Billion, 2022
Figure 84: Global Multicancer Screening Market (by Region), $Billion, 2021-2032
Figure 85: North America Multicancer Screening Market, $Billion, 2021-2032
Figure 86: North America Multicancer Screening Market (by Country), $Billion, 2021-2032
Figure 87: U.S. Multicancer Screening Market, $Billion, 2021-2032
Figure 88: Canada Multicancer Screening Market, $Billion, 2021-2032
Figure 89: Europe Multicancer Screening Market, $Billion, 2021-2032
Figure 90: Europe Multicancer Screening Market (by Country), $Million, 2021-2032
Figure 91: Germany Multicancer Screening Market, $Million, 2021-2032
Figure 92: France Multicancer Screening Market, $Million, 2021-2032
Figure 93: U.K. Multicancer Screening Market, $Million, 2021-2032
Figure 94: Italy Multicancer Screening Market, $Million, 2021-2032
Figure 95: Spain Multicancer Screening Market, $Million, 2021-2032
Figure 96: Rest-of-Europe Multicancer Screening Market, $Million, 2021-2032
Figure 97: Asia-Pacific Multicancer Screening Market, $Billion, 2021-2032
Figure 98: Asia-Pacific Multicancer Screening Market Share (by Country), $Million, 2021-2032
Figure 99: China Multicancer Screening Market, $Million, 2021-2032
Figure 100: Japan Multicancer Screening Market, $Million, 2021-2032
Figure 101: India Multicancer Screening Market, $Million, 2021-2032
Figure 102: Australia Multicancer Screening Market, $Million, 2021-2032
Figure 103: South Korea Multicancer Screening Market, $Million, 2021-2032
Figure 104: Rest-of-Asia-Pacific Multicancer Screening Market, $Million, 2021-2032
Figure 105: Latin America Multicancer Screening Market, $Million, 2021-2032
Figure 106: Latin America Multicancer Screening Market Share (by Country), $Million, 2021 and 2032
Figure 107: Brazil Multicancer Screening Market, $Million, 2021-2032
Figure 108: Mexico Multicancer Screening Market, $Million, 2021-2032
Figure 109: Rest-of-Latin America Multicancer Screening Market, $Million, 2021-2032
Figure 110: Rest-of-the-World Multicancer Screening Market, $Million, 2021-2032
Figure 111: Global Multicancer Screening Market, Total Number of Companies Profiled
Figure 112: Global Multicancer Screening Market: Value Chain
Figure 113: Agilent Technologies, Inc.: Product Portfolio
Figure 114: Agilent Technologies, Inc.: Overall Financials, $Million, 2020-2022
Figure 115: Agilent Technologies, Inc.: Revenue (by Segment), $Million, 2020-2022
Figure 116: Agilent Technologies, Inc.: Revenue (by Region), $Million, 2020-2022
Figure 117: Agilent Technologies, Inc.: R&D Expenditure, $Million, 2020-2022
Figure 118: Atara Biotherapeutics, Inc.: Product Portfolio
Figure 119: Atara Biotherapeutics, Inc.: Overall Financials, $Million, 2020-2022
Figure 120: Atara Biotherapeutics, Inc.: R&D Expenditure, $Million, 2020-2022
Figure 121: Burning Rock DX: Product Portfolio
Figure 122: Danaher Corporation (Integrated DNA Technologies, Inc.): Product Portfolio
Figure 123: Danaher Corporation (Integrated DNA Technologies, Inc.): Overall Financials, $Million, 2020-2022
Figure 124: Danaher Corporation (Integrated DNA Technologies, Inc.): Revenue (by Segment), $Million, 2020-2022
Figure 125: Danaher Corporation (Integrated DNA Technologies, Inc.): Revenue (by Region), $Million, 2020-2022
Figure 126: Danaher Corporation (Integrated DNA Technologies, Inc.): R&D Expenditure, $Million, 2020-2022
Figure 127: Exact Sciences Corporation: Product Portfolio
Figure 128: Exact Sciences Corporation: Overall Financials, $Million, 2019-2021
Figure 129: Exact Sciences Corporation: Revenue (by Segment), $Million, 2019-2021
Figure 130: Exact Sciences Corporation: Revenue (by Region), $Million, 2019-2021
Figure 131: Exact Sciences Corporation: R&D Expenditure, $Million, 2019-2021
Figure 132: F. Hoffmann-La Roche Ltd: Product Portfolio
Figure 133: F. Hoffmann-La Roche Ltd: Overall Financials, 2020-2022
Figure 134: F. Hoffmann-La Roche Ltd: Revenue (by Segment), 2020-2022
Figure 135: F. Hoffmann-La Roche Ltd: R&D Expenditure, 2020-2022
Figure 136: Fulgent Genetics.: Product Portfolio
Figure 137: Fulgent Genetics.: Overall Financials, 2020-2022
Figure 138: Fulgent Genetics.: Revenue (by Region), 2020-2022
Figure 139: Fulgent Genetics.: R&D Expenditure, 2020-2022
Figure 140: Genecast Biotechnology Co., Ltd: Product Portfolio
Figure 141: Guardant Health, Inc.: Product Portfolio
Figure 142: Guardant Health, Inc.: Overall Financials, $Million, 2019-2021
Figure 143: Guardant Health, Inc.: Revenue (by Segment), $Million, 2019-2021
Figure 144: Guardant Health, Inc.: Revenue (by Region), $Million, 2019-2021
Figure 145: Guardant Health, Inc.: R&D Expenditure, $Million, 2019-2021
Figure 146: Illumina, Inc. (GRAIL, LLC.): Product Portfolio
Figure 147: Illumina, Inc. (GRAIL, LLC.): Overall Financials, $Million, 2020-2022
Figure 148: Illumina, Inc. (GRAIL, LLC): Revenue (by Segment), $Million, 2020-2022
Figure 149: Illumina, Inc. (GRAIL, LLC.): Revenue (by Region), $Million, 2020-2022
Figure 150: Illumina, Inc. (GRAIL, LLC.): R&D Expenditure, $Million, 2020-2022
Figure 151: Konica Minolta, Inc. (Ambry Genetics.): Product Portfolio
Figure 152: Konica Minolta, Inc. (Ambry Genetics.): Overall Financials, $Million, 2019-2021
Figure 153: Konica Minolta, Inc. (Ambry Genetics.): Revenue (by Segment), $Million, 2019-2021
Figure 154: Konica Minolta, Inc. (Ambry Genetics.): Revenue (by Region), $Million, 2019-2021
Figure 155: Konica Minolta, Inc. (Ambry Genetics.): R&D Expenditure, $Million, 2019-2021
Figure 156: Laboratory Corporation of America Holdings.: Product Portfolio
Figure 157: Laboratory Corporation of America Holdings.: Overall Financials, $Million, 2020-2022
Figure 158: Laboratory Corporation of America Holdings.: Revenue (by Segment), $Million, 2020-2022
Figure 159: Laboratory Corporation of America Holdings.: Revenue (by Region), $Million, 2020-2022
Figure 160: Myriad Genetics, Inc.: Product Portfolio
Figure 161: Myriad Genetics, Inc.: Overall Financials, ($Million), 2020-2022
Figure 162: Myriad Genetics, Inc.: Revenue (by Segment), $Million, 2020-2022
Figure 163: Myriad Genetics, Inc.: R&D Expenditure, ($Million), 2020-2022
Figure 164: Tempus: Product Portfolio
Figure 165: Thermo Fisher Scientific Inc.: Product Portfolio
Figure 166: Thermo Fisher Scientific Inc.: Overall Financials, $Million, 2020-2022
Figure 167: Thermo Fisher Scientific Inc.: Revenue (by Segment), $Million, 2020-2022
Figure 168: Thermo Fisher Scientific Inc.: Revenue (by Region), $Million, 2020-2022
Figure 169: Thermo Fisher Scientific Inc.: R&D Expenditure, $Million, 2020-2022
Market Report Coverage
|
Multicancer Screening Market |
|||
|
Base Year |
2022 |
Market Size in 2022 |
$1.90 Billion |
|
Forecast Period |
2023-2032 |
Value Projection and Estimation by 2032 |
$7.78 Billion |
|
CAGR During Forecast Period |
15.09% |
Number of Tables |
13 |
|
Number of Pages |
228 |
Number of Figures |
169 |
Key Market Players and Competition Synopsis
The companies that are profiled have been selected based on inputs gathered from primary experts, analyzing company coverage, product portfolio, and market penetration.
The top segment players leading the market include in-vitro diagnostics (IVDs) and laboratory developed tests (LDTs). IVD kit providers constitute around 30.54% of the presence in the market, and LDT kit providers constitute 69.46% of the multicancer screening market.
Key Companies Profiled
• Agilent Technologies, Inc.
• Atara Biotherapeutics, Inc.
• Burning Rock DX
• Danaher Corporation
• Exact Sciences Corporation
• F. Hoffmann-La Roche Ltd
• Fulgent Genetics.
• Genecast Biotechnology Co., Ltd
• Guardant Health, Inc.
• Illumina, Inc.
• Konica Minolta, Inc.
• Laboratory Corporation of America Holdings.
• Myriad Genetics, Inc.
• Tempus
• Thermo Fisher Scientific Inc.
How can this report add value to an organization?
Product/Innovation Strategy: The report considers multicancer screening kit/panel and assay product-based companies. The industry is seeing constant development and product launches with new and innovative upgrades. Additionally, new discoveries of specific biomarkers related to various cancer types are increasing researchers' trust in multicancer sequencing.
Growth/Marketing Strategy: The key components in multicancer screening are the kits and the related technologies for sample analysis. The advancements in new analysis methods like liquid biopsy are influencing the growth of this market. Additionally, the discovery of biomarkers and other genes for various diseases is helping panel manufacturers curate precise kits and assays for multicancer screening of various cancer types.
Competitive Strategy: The key players in the global multicancer screening market have been analyzed and profiled in the study, consisting of most product-based companies, as well as a few emerging companies. Moreover, a detailed competitive benchmarking of the players operating in the global multicancer screening market has been done to help the reader understand how players stack against each other, presenting a clear market landscape. Additionally, comprehensive competitive strategies such as partnerships, agreements, and collaborations will aid the reader in understanding the untapped revenue pockets in the market.
Multicancer Screening Industry and Technology Overview
The global multicancer screening market is projected to reach $7.78 billion by 2032 from $1.90 billion in 2022, growing at a CAGR of 15.09% during the forecast period 2023-2032. The growth in the global multicancer screening market is expected to be driven by the increased use of liquid biopsy testing and increasing prevalence of cancers.
Market Lifecycle Stage
Multicancer screening is a cancer screening test that has the potential to screen for more than one cancer type with a single test. This test can be tissue-based or liquid biopsy-based. Most of the newly developed multicancer screening tests in the market right now are liquid biopsy-based, which detects cancer biomarkers in bodily fluids such as blood, saliva, urine, etc. Biomarkers/genetic markers from RNA, DNA, or protein are analyzed from these samples. If any of these biomarkers or gene mutations are positive in the analysis, further follow-up tests need to be performed to confirm the findings.
Some multicancer screening tests can also identify the tissue of the origin of cancer, while others need imaging tests to verify the origin. There are a lot of laboratory-based multicancer screening tests available in the market right now, and a few have also received U.S. Food and Drug Administration (FDA) and conformité européenne (CE) approvals as well. Multicancer screening tests are also important because, according to the America Cancer Society, Inc., 70% of all cancer deaths are from cancers that still do not have any proven individual screening tests. This can be extremely decreased with the advent of new biomarkers through increasing funding investments in multicancer screening tests.
Figure: Global Multicancer Screening Market Snapshot
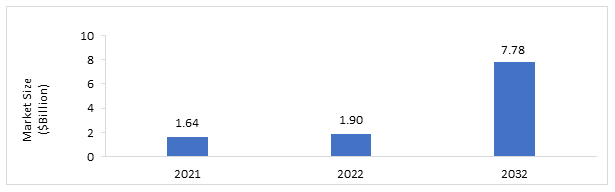
Source: BIS Research Analysis
Impact
• For instance, in October 2020, Exact Sciences Corporation launched Oncotype MAP Pan-Cancer Tissue test for patients with metastatic, advanced, recurrent, or refractory cancer. The Oncoptype MAP test provides clinically actionable information from hundreds of cancer-related genes that allows physicians to understand the tumor profile of patients and recommend effective clinical trials or targeted therapies.
• In February 2022, GRAIL, LLC. (Illumina, Inc.) collaborated with Point32Health for a two-phased pilot trial of Galleri, which is GRAIL's proprietary multicancer blood test. With this collaboration, Point32Health is the first health plan that would work with GRAIL to complement the recommended cancer screenings.
Impact of COVID-19
In 2019, the world was hit by a pandemic. The culprit is coronavirus disease (COVID-19), which is an illness caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) virus. SARS-CoV-2 was found to be a novel type of coronavirus that started causing severe illness and deaths around the world. The global pandemic began in early 2020, causing mild respiratory tract infections to serious lower respiratory infections. The first case of coronavirus was reported in Wuhan, China, in December 2019.
According to an electronic medical record company, there was approximately an 80%-90% decline in screenings of breast, cervical, and colorectal cancers in patients during March and April of 2020 as compared to the same months in 2019. Even though screenings did increase in June 2020, they were still down by 29%-36%. Additionally, according to the Centers for Disease Control and Prevention, HPV vaccinations dropped by 73% between February and April 2020. The full impact of these delays will be realized in the coming years.
Market Segmentation:
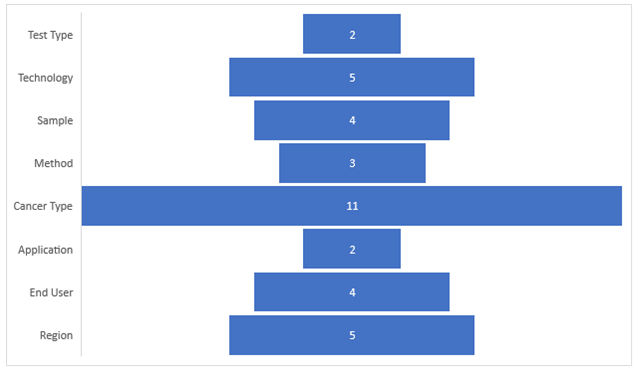
Segmentation 1: by Test Type
• Laboratory Developed Tests (LDTs)
• In-Vitro Diagnostics (IVDs)
The global multicancer screening market (by test type) is expected to be dominated by the laboratory developed tests (LDTs) segment during the forecast period 2023-2032. The in-vitro diagnostics (IVDs) segment is expected to grow with a high CAGR and have a market share of 35.72% till 2032.
Segmentation 2: by Technology
• Next-Generation Sequencing (NGS)
• Polymerase Chain Reaction (PCR)
• Immunohistochemistry (IHC)
• Fluorescence In-Situ Hybridization (FISH)
• Other Technologies
The global multicancer screening market (by technology) is expected to be dominated by the next-generation sequencing (NGS) segment during the forecast period 2023-2032. This is because it is the most efficient and accurate technology currently available when it comes to gene analysis.
Segmentation 3: by Sample
• Tissue
• Blood
• Saliva and Buccal Swab
• Others
The global multicancer screening market (by sample) is expected to be dominated by the tissue segment during the forecast period 2023-2032. This is due to the high validation and trust on these tests as compared to the newer liquid biopsy-based tests.
Segmentation 4: by Method
• DNA
• RNA
• Proteins
The DNA segment is expected to dominate the global multicancer screening market (by method) during the forecast period 2023-2032. This growth in this segment is attributed to the fact that DNA analysis is the most convenient, even with the fast-growing liquid biopsy methods.
Segmentation 5: by Cancer Type
• Breast and Gynecologic
• Gastrointestinal
• Endocrine
• Genitourinary
• Skin
• Brain/Nervous System
• Sarcoma
• Hematological Malignancies
• Lung
• Head and Neck
• Other Cancer Types
The global multicancer screening market (by cancer type) is expected to be dominated by the breast and gynecological segment during the forecast period 2023-2032. This is because the high prevalence of these cancer types and high number of known biomarkers makes them easy to screen for.
Segmentation 6: by Application
• Clinical
• Research
As of 2021, the global multicancer screening market (by application) was dominated by the research segment, holding a 85.06% market share.
Segmentation 7: by End User
• Hospitals
• Diagnostic and Clinical Laboratories
• Academic and Research Institutions
• Other End Users
As of 2021, the global multicancer screening market (by end user) was dominated by the academic and research institutions segment, holding a 62.17% market share.
Segmentation 8: by Region
• North America
• Europe
• Asia-Pacific
• Latin America
• Rest-of-the-World
North America is expected to dominate the global multicancer screening market with a revenue of $0.87 billion in 2021. However, the Asia-Pacific (APAC) region, constituting several emerging economies, is expected to register the highest CAGR of 17.60% during the forecast period 2023-2032.
Recent Developments in Global Multicancer Screening Market
• In March 2021, Agilent Technologies, Inc. made an agreement to acquire Resolution Bioscience Inc. to expand the company's NGS-based cancer diagnostics and its fast-growing precision medicine segment. This agreement was made for $550 million in cash at closing and an additional $145 million when the company achieves future performance milestones.
• In December 2022, Integrated DNA Technologies, Inc. acquired Invitae Corporation.'s next-generation sequencing (NGS) research assays with the Archer trademark for $48 million.
• In January 2023, Laboratory Corporation of America Holdings. acquired Personal Genome Diagnostics for $450 million and for another $125 million as contingent milestone payments. Personal Genome Diagnostics was a next-generation sequencing test maker, and with this acquisition, Laboratory Corporation of America Holdings. has increased its NGS testing portfolio.
• In January 2023, FOUNDATION MEDICINE, INC. (F. Hoffmann-La Roche Ltd) got FDA approval for its FoundationOne Liquid CDx as a companion diagnostic for Rozlytrek. This test is relevant for ROS1+NSCLC patients and NTRK fusion-positive solid tumors patients.
• In January 2023, Burning Rock DX got an FDA Breakthrough Device Designation for its Multi-Cancer Detection Blood Test called OverC. This test is intended for the detection of lung, liver, ovarian, esophageal, and pancreatic cancer for adults aged between 50 and 75 years.
Demand – Drivers and Limitations
Following are the demand drivers for the global multicancer screening market:
• Early Detection of Cancer Leads to Higher Survival Chances and Better Treatment
• Increasing Popularity of Liquid Biopsy Leading to Easier Screening
• Reduced Treatment Cost Due to Earlier Cancer Detection
The market is expected to face some limitations too due to the following challenges:
• Lack of Awareness about the Multicancer Screening Tests
• Too Costly to be Used as Preventive Care
Analyst’s View
According to Swati Sood, Principal Analyst, BIS Research, “Global multicancer screening market is a rapidly emerging market and is expected to revolutionize the field of oncology field in the coming years due to the continuous increase in the demand for cancer diagnosis and industrial analysis.”
Multicancer Screening Market - A Global and Regional Analysis
Focus on Test Type, Technology, Sample, Method, Cancer Type, Application, End User, and Region Analysis - Analysis and Forecast, 2023-2032
Frequently Asked Questions
Multicancer screening is a cancer screening test that has the potential to screen for more than one cancer type with a single test. This test can be tissue-based or liquid biopsy-based. Most of the newly developed multicancer screening tests in the market right now are liquid biopsy-based, which detect cancer biomarkers in bodily fluids such as blood, saliva, urine, etc. Biomarkers/genetic markers from RNA, DNA, or protein are analyzed from these samples. If any of these biomarkers or gene mutations are positive in the analysis, further follow-up tests need to be performed to confirm the findings.
The major advantage of multicancer screening, as compared to individual screening tests, is the ability to screen for multiple tests at the same time. The increasing prevalence of cancer due to the changing lifestyle around the globe calls for a major need for screening tests. Additionally, the advancements in liquid biopsy-based methods and better analysis using technologies like NGS are really increasing the market potential and are expected to revolutionize the oncology field in the coming years.
The global multicancer screening market has seen major developments such as business expansions, partnerships, collaborations, and joint ventures by key players operating in the market. According to BIS Research analysis, the majority of the companies preferred partnerships, collaborations, and joint ventures in addition to multiple product launches as a strategy to further increase their growth in the global multicancer screening market.
A new entrant needs to make sure that the different cancers being tested for are using a validated analytical method, and the discovery of new biomarkers is also an advantage. Additionally, the ability to screen a few rare cancer types can be an advantage. Furthermore, approvals from FDA are still hard to get, and thus all the necessary precautions should be taken to be able to launch it as an LDT as well.
The following can be considered the USPs of the report:
· Vast segmentation of the report, especially the vast number of cancer types considered
· Product specifications and comparative analysis
· Market dynamic analysis of the opportunities, trends, and challenges in the market
· Competitive benchmarking of key players for each product type
· Growth share analysis for key segments
This report will be useful for firms planning to enter the multicancer screening market and investors who want to invest in the product. Additionally, established market players in this market can also benefit from this report. This is because the report discusses the benefits of using multicancer screening tests and the latest advancements in the market right now.




